Are you curious about the fascinating world of chemistry and looking to uncover the secrets of diatomic elements? You’re in the right place! Diatomic elements, the simplest yet most intriguing molecules, form the backbone of many chemical reactions and natural processes. In this comprehensive guide, we'll dive deep into what makes these elements unique, how they behave in various conditions, and how you can practically apply your newfound knowledge.
Why Understanding Diatomic Elements Matters
Understanding diatomic elements is more than just a scientific curiosity—it’s essential for anyone interested in chemistry. Diatomic elements consist of just two atoms bonded together, such as hydrogen (H2), oxygen (O2), and nitrogen (N2). These elements play a crucial role in everyday life, from the air we breathe to the fuel that powers our vehicles. Mastery over these fundamental concepts can provide you with the tools to tackle complex chemical problems, whether in a laboratory setting or in real-world applications.
Immediate Action: Kickstart Your Knowledge
To start, it’s important to grasp some foundational concepts quickly. Here’s what you should focus on first:
Quick Reference
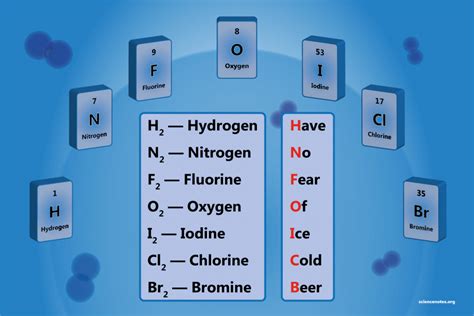
- Immediate action item: Identify the seven diatomic elements: H2, N2, O2, F2, Cl2, Br2, and I2. Understanding these elements will form the basis of your knowledge.
- Essential tip: Learn the common physical and chemical properties of these elements. For instance, molecular oxygen (O2) is a colorless, odorless gas, essential for respiration in living organisms.
- Common mistake to avoid: Confusing diatomic elements with diatomic compounds like water (H2O) or carbon dioxide (CO2). Remember, these are different and should not be interchanged.
Detailed Guide to Hydrogen (H2)
Hydrogen, the simplest element with one proton and one electron, exists as H2 under standard conditions. It’s a colorless, odorless, and highly flammable gas. Hydrogen is the most abundant element in the universe and has numerous applications.
Basic Properties and Uses
Hydrogen makes up about 75% of the universe’s elemental mass. It’s vital for various industrial processes:
- Rocket fuel: Liquid hydrogen is a key component of rocket fuel due to its high energy content.
- Hydrogenation: It’s used in converting unsaturated fats to saturated fats by adding hydrogen atoms.
- Acids: The hydrogen ion (H+) is central to acids, which are fundamental to many chemical reactions.
How to Observe and Measure Hydrogen
Handling hydrogen requires caution due to its flammability. Here’s how to safely observe and measure it:
- Store hydrogen gas in properly rated cylinders and always follow safety protocols.
- To measure hydrogen, use a gas analyzer designed for flammable gases.
- Conduct experiments in a well-ventilated area or a fume hood to prevent any potential build-up of gas.
Best Practices in Using Hydrogen
For those working with hydrogen in industrial or research settings:
- Always use appropriate personal protective equipment (PPE).
- Install explosion-proof equipment to mitigate risks.
- Regularly inspect and maintain all equipment used with hydrogen to prevent leaks.
Detailed Guide to Nitrogen (N2)
Nitrogen, making up about 78% of Earth’s atmosphere, is a major diatomic element (N2). It’s colorless, odorless, and generally inert under standard conditions. This element is crucial for life and various industrial processes.
Properties and Applications
Nitrogen plays a vital role in:
- Amino Acid Synthesis: It’s a fundamental component in the formation of amino acids, essential for protein synthesis in living organisms.
- Fertilizers: Nitrogen in the form of ammonia (NH3) is a key ingredient in many fertilizers.
- Cryogenics: Liquid nitrogen is used in cryogenics due to its extremely low boiling point of -196°C.
Handling Nitrogen Safely
Though nitrogen itself is inert, it can displace oxygen in confined spaces, leading to asphyxiation:
- Ensure proper ventilation when handling gaseous nitrogen.
- Use gas detectors to monitor nitrogen levels and avoid enclosed spaces.
- Train personnel in safe handling and emergency procedures for nitrogen-related incidents.
Industrial Uses of Nitrogen
Here’s how nitrogen is put to practical use in industry:
- Atmospheric Control: Nitrogen is used to purge air from vacuum chambers and control the atmosphere in metallurgical processes.
- Food Preservation: It’s used in packaging to extend the shelf life of perishable goods.
- Manufacturing: Nitrogen is a key component in producing semiconductor devices and other precision instruments.
Practical FAQ: Diatomic Elements Explained
What are the physical states of diatomic elements at room temperature?
At room temperature, the diatomic elements exist in different states based on their boiling and melting points. Here’s a quick overview:
| Element | State at Room Temperature |
|---|---|
| Hydrogen (H2) | Gas |
| Nitrogen (N2) | Gas |
| Oxygen (O2) | Gas |
| Fluorine (F2) | Gas |
| Chlorine (Cl2) | Gas |
| Bromine (Br2) | Liquid |
| Iodine (I2) | Solid |
Conclusion: Mastery of Diatomic Elements
Understanding the basics and practical applications of diatomic elements will not only enhance your knowledge but also equip you with the ability to implement these principles in various fields. From laboratory experiments to industrial applications, the knowledge of hydrogen, nitrogen, and other diatomic elements is crucial. With this guide, you’ve been provided with actionable steps, practical tips, and detailed insights to navigate and utilize your newfound knowledge effectively.