Acetylene is a fundamental molecule in organic chemistry, primarily recognized for its simple yet complex structure. This guide is designed for those who need to understand how to draw and interpret the acetylene (C2H2) Lewis structure. This step-by-by-step guidance will provide you with actionable advice on creating the Lewis structure and address the common pain points you might encounter.
Problem-Solution Opening Addressing User Needs
If you’ve ever struggled to grasp the fundamentals of acetylene’s Lewis structure, you’re not alone. Many students and even seasoned chemists can find themselves tangled in the specifics of valence electrons, bonding, and the geometry involved. Understanding this structure is crucial for grasping more advanced concepts in chemistry, like molecular orbital theory and reactivity patterns. This guide is designed to demystify the Lewis structure of acetylene, offering clear, straightforward, and practical advice to help you build an accurate representation and understand its significance.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Start by drawing a single bond between the two carbon atoms.
- Essential tip with step-by-step guidance: Place four hydrogen atoms, two on each carbon atom to fulfill the octet rule.
- Common mistake to avoid with solution: Avoid forgetting that the two carbon atoms share three bonds (a triple bond) instead of just one or two.
How to Draw the Acetylene (C2H2) Lewis Structure
Drawing the acetylene Lewis structure can seem daunting, but breaking it down into manageable steps makes it achievable. Here’s how you can approach it:
Step 1: Count the total number of valence electrons.
Acetylene has two carbon atoms and two hydrogen atoms. Carbon has 4 valence electrons, and hydrogen has 1. Thus, we have:
2 (carbons) × 4 (valence electrons per carbon) + 2 (hydrogens) × 1 (valence electron per hydrogen) = 10 valence electrons.
Step 2: Place the least electronegative atoms in the center.
In acetylene, carbon is less electronegative than hydrogen, so the two carbon atoms are placed in the center:
Step 3: Connect outer atoms to the central atom with single bonds and distribute the remaining electrons.
Connect each carbon atom to a hydrogen atom with a single bond:
H - C - C - H.
This uses up 4 of the 10 valence electrons, leaving 6 electrons to distribute:
Step 4: Complete the octet rule for each atom.
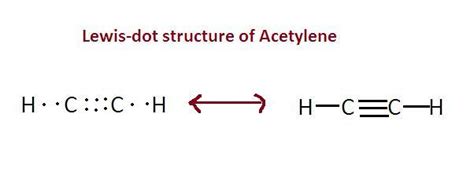
Carbon needs 8 electrons to fulfill the octet rule, and hydrogen needs only 2 (it’s already satisfied). To fulfill the octet rule for carbons, we need to add three bonds between the two carbon atoms:
This creates a triple bond and uses up all 10 valence electrons:
· · H - C ≡ C - H · ·
Step 5: Check the structure to ensure all atoms have a full octet.
Each carbon now has 8 electrons (4 from bonds and 4 non-bonding electrons), and each hydrogen has 2 electrons:
Final Acetylene Lewis Structure:
· · H - C ≡ C - H · ·
This triple bond structure is crucial for understanding the properties and reactivity of acetylene.
Practical FAQ
Why does acetylene have a triple bond?
Acetylene (C2H2) has a triple bond between the two carbon atoms because each carbon atom needs to share electrons to fulfill their octet rule. In a triple bond, three pairs of electrons are shared between the two carbons, allowing each to achieve a stable electron configuration with 8 electrons in its valence shell.
What are common errors to avoid when drawing the acetylene Lewis structure?
Common errors include:
- Incorrect valence electron count: Ensure you correctly count the valence electrons from all atoms.
- Mistake in bond type: Remember, acetylene has a triple bond between the carbon atoms, not just a single or double bond.
- Hydrogen placement: Place each hydrogen on a separate carbon atom and use single bonds, not double or triple bonds.
Avoiding these mistakes will help ensure your Lewis structure is accurate.
By following this guide, you’ll be able to construct the acetylene Lewis structure with confidence and clarity, paving the way for a deeper understanding of this essential molecule in organic chemistry.