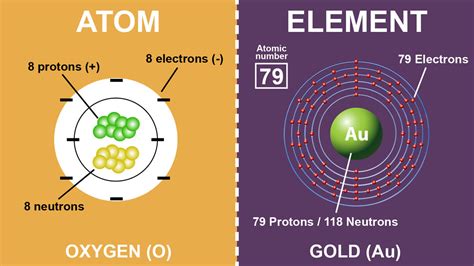
All atoms of an element possess a unique and fascinating characteristic: they have the same number of protons. This defining attribute, known as the atomic number, distinguishes one element from another on the periodic table and forms the foundation of atomic theory. This inherent sameness within elements provides a key insight into the behavior of matter at a microscopic level. Understanding this concept not only advances scientific discovery but also has practical implications in fields ranging from chemistry to medicine.
Key Insights
- Primary insight with practical relevance: All atoms of an element have the same atomic number, which determines the element’s identity and chemical properties.
- Technical consideration with clear application: This principle underpins many scientific methods, such as isotopic analysis and radioactive dating.
- Actionable recommendation: To understand the behavior of an element, focus on its atomic number and resultant electron configuration.
Atomic Structure and the Concept of Protons
At the heart of every element is its atomic structure, primarily characterized by protons, neutrons, and electrons. Protons are positively charged particles found in the nucleus of an atom, and they define the element’s atomic number. For instance, carbon atoms always have six protons, which makes them unique from oxygen atoms that have eight protons. This constancy in proton number ensures that each element exhibits predictable chemical behavior based on its electron arrangement.The Significance of Atomic Number in Chemistry
The atomic number holds immense significance in chemistry because it directly correlates to an element’s position on the periodic table and influences its chemical reactivity. Elements with the same number of protons will have similar chemical properties. For example, all alkali metals—such as lithium, sodium, and potassium—share a single valence electron, which leads to their characteristic high reactivity and similar chemical behaviors. Understanding atomic number provides crucial insights into chemical bonding, molecular structure, and the overall properties of elements and compounds.What determines the identity of an element?
The atomic number, which represents the number of protons in the nucleus of an atom, determines the identity of an element. Each element has a unique atomic number, establishing its place on the periodic table.
How do isotopes affect the properties of an element?
Isotopes are variants of an element that have the same number of protons but different numbers of neutrons. While isotopes have identical chemical properties due to the same atomic number, they can have different physical properties, such as mass and stability, which influence applications in fields like medicine and nuclear physics.
In summary, the concept that all atoms of an element have the same number of protons, encapsulated in the atomic number, is a foundational principle that governs the behavior of elements and compounds. This simple yet profound truth allows scientists to predict and understand the vast array of chemical phenomena, contributing to advancements in numerous scientific and technological fields.