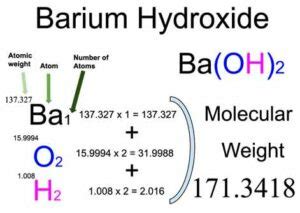
Welcome to our comprehensive guide on calculating molar mass—a crucial concept in chemistry that will help you unlock a deeper understanding of chemical reactions and molecular structures. Whether you are a student preparing for exams, a professional seeking precise calculations, or a curious mind interested in the finer details of chemistry, this guide is designed to meet your needs. Let's dive in and demystify molar mass calculation step by step.
Understanding the Problem: Why Molar Mass Matters
Molar mass is the weight of one mole of a substance, measured in grams per mole (g/mol). It’s fundamental in understanding how substances interact in chemical reactions. Knowing the molar mass allows chemists to precisely measure how much of a substance they need, predict reaction yields, and more. However, for many, accurately calculating molar mass can be tricky, often leading to errors that can compound in complex reactions.
Let’s address the common challenges. Many learners struggle with differentiating between atomic mass and molar mass, finding it hard to identify the periodic table elements’ correct atomic weights for calculation. Additionally, they may face difficulties managing compounds that contain polyatomic ions, leading to confusion in determining the correct molar mass.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Always double-check the periodic table for the most accurate atomic weights before calculating molar mass.
- Essential tip with step-by-step guidance: Break down complex compounds by identifying the individual elements and their quantities.
- Common mistake to avoid with solution: Avoid rounding atomic weights too early; wait until the final calculation for the most precise result.
How to Calculate Molar Mass of Simple Compounds
Let’s start with the basics: calculating the molar mass of simple, binary compounds—those made up of two elements. Follow this straightforward process:
- Identify the elements: List the elements present in the compound. For example, consider water (H2O).
- Look up atomic masses: Use a reliable periodic table to find the atomic masses of hydrogen (H) and oxygen (O).
- Calculate molar mass: Multiply the atomic mass of each element by the number of its atoms in the compound. For H2O, multiply the atomic mass of hydrogen (1.008 g/mol) by 2, and multiply the atomic mass of oxygen (16.00 g/mol) by 1. Add these products together: (2 * 1.008) + 16.00 = 18.016 g/mol.
Through this process, you determine the molar mass of water to be approximately 18.016 g/mol.
How to Calculate Molar Mass of Complex Compounds
Complex compounds, including those with polyatomic ions or multiple elements, require a slightly more intricate approach. Let’s break it down step-by-step:
- Identify and list components: Identify the elements and polyatomic ions present in the compound. For example, consider calcium carbonate (CaCO3).
- Break down the compound: Identify each element and its respective quantity in the formula.
- Calculate atomic masses: Find the atomic masses for calcium (Ca), carbon (C), and oxygen (O) on the periodic table. Note: Oxygen appears three times in CaCO3.
- Perform calculations: Multiply each element's atomic mass by its quantity and add the results. For CaCO3: Ca (40.08 g/mol) x 1 + C (12.01 g/mol) x 1 + O (16.00 g/mol) x 3 = 40.08 + 12.01 + 48.00 = 100.09 g/mol.
Thus, the molar mass of calcium carbonate is approximately 100.09 g/mol.
Tips and Best Practices for Accurate Molar Mass Calculation
Precision in molar mass calculation is key to success in chemistry. Here are some best practices:
- Use accurate atomic masses: Always use the most current atomic mass values from a reliable source like the IUPAC periodic table.
- Be meticulous with stoichiometry: Ensure that the number of each element’s atoms in the formula is accurate.
- Avoid premature rounding: Do not round intermediate results. Round only your final answer to maintain precision.
Common Mistakes and How to Avoid Them
Even experienced chemists can make mistakes when calculating molar mass. Here are common pitfalls and how to avoid them:
- Mistake: Misidentifying polyatomic ions: Polyatomic ions like nitrate (NO3−) have fixed formulas. Always refer to a list of common polyatomic ions to avoid errors.
- Mistake: Incorrect use of subscripts: Subscripts indicate the number of atoms in a molecule. Ensure you correctly apply these in your calculations.
- Mistake: Using outdated atomic masses: Atomic masses can vary slightly depending on the isotope composition of naturally occurring elements. Use the most recent values available.
Practical FAQ
What is the molar mass of sulfuric acid (H2SO4)?
To find the molar mass of sulfuric acid (H2SO4), follow these steps:
- Identify the elements in the compound: H, S, and O.
- Look up atomic masses: H (1.008 g/mol), S (32.07 g/mol), O (16.00 g/mol).
- Calculate the molar mass: H2 (2 * 1.008) + S (1 * 32.07) + O4 (4 * 16.00) = 2.016 + 32.07 + 64.00 = 98.086 g/mol.
The molar mass of sulfuric acid (H2SO4) is approximately 98.086 g/mol.
Through these detailed steps and practical tips, you'll gain the confidence to tackle molar mass calculations accurately. Remember, practice makes perfect—apply these methods to different compounds and continually refine your approach.
This guide aims to equip you with the knowledge and confidence to dive deeper into chemistry, ensuring precision and understanding in your studies or professional endeavors. Happy calculating!