The bicarbonate buffer system is a critical component of the body’s internal homeostasis, functioning seamlessly to maintain pH balance in the extracellular fluids. Often hailed as nature’s pH secret, it plays an essential role in sustaining the delicate equilibrium of our bodily functions. The intricate balance facilitated by this buffer system is paramount in preventing the catastrophic shifts that would occur in enzyme activity, nerve transmission, and muscle contraction without it.
Understanding the Bicarbonate Buffer System
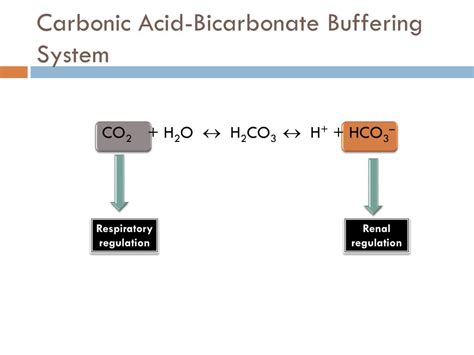
At its core, the bicarbonate buffer system involves a dynamic equilibrium between carbon dioxide (CO2) and bicarbonate (HCO3-) ions. This buffer mechanism operates through the reversible reaction: CO2 + H2O ⇌ H2CO3 ⇌ H+ + HCO3-. Here, carbon dioxide, a byproduct of cellular metabolism, reacts with water to form carbonic acid, which quickly dissociates into hydrogen and bicarbonate ions. When acidity rises, bicarbonate ions combine with excess hydrogen ions to form carbonic acid, which is subsequently converted into CO2 and expelled via the respiratory system, thus lowering the pH. Conversely, when alkalinity increases, CO2 is formed from bicarbonate, reducing the pH.
Key Insights
- Primary insight with practical relevance: The bicarbonate buffer system ensures the pH level remains stable, thus maintaining normal physiological functions.
- Technical consideration with clear application: Understanding the relationship between CO2 and bicarbonate can help in diagnosing and managing metabolic acidosis and alkalosis.
- Actionable recommendation: Monitoring blood gas levels is crucial for effective treatment of acid-base imbalances.
Physiological Significance
The physiological significance of the bicarbonate buffer system cannot be overstated. This system is particularly crucial in the bloodstream, where it works in tandem with the respiratory system to buffer excess hydrogen ions and maintain a pH range of about 7.35 to 7.45. Deviations outside this narrow range can have severe implications, influencing metabolic processes and cellular functions. The bicarbonate buffer system’s ability to act swiftly and efficiently ensures that the body's internal environment remains conducive to life. Moreover, it plays a pivotal role in the regulation of blood pH, making it indispensable in conditions such as respiratory and metabolic diseases.
Clinical Implications
In clinical practice, the bicarbonate buffer system’s understanding is vital for managing acid-base disorders. For instance, in metabolic acidosis, there is an accumulation of acids or a relative loss of bicarbonate, resulting in decreased blood pH. On the other hand, metabolic alkalosis occurs due to an excess of bicarbonate or a loss of acid. Clinical intervention often involves adjusting bicarbonate levels to correct these imbalances. For example, in patients with chronic respiratory failure, administering bicarbonate can help manage metabolic acidosis by compensating for the reduced CO2 excretion through respiration. Thus, a comprehensive grasp of the bicarbonate buffer system is essential for accurate diagnosis and effective treatment.
What role does the bicarbonate buffer system play in respiration?
The bicarbonate buffer system works in conjunction with the respiratory system to regulate blood pH. When CO2 levels increase, the bicarbonate ion absorbs excess hydrogen ions, preventing a significant drop in pH. Simultaneously, increased CO2 is expelled via the lungs, helping to restore the pH balance.
Can dietary changes affect the bicarbonate buffer system?
Yes, dietary changes can affect the bicarbonate buffer system. Diets high in acidic foods can lead to an excess of hydrogen ions, potentially disrupting the buffer system's equilibrium. Conversely, diets rich in alkaline substances like fruits and vegetables can help maintain a balanced pH level.
The bicarbonate buffer system’s importance extends beyond physiological boundaries into clinical realms where understanding and managing this intricate balance is essential for effective medical interventions. With its dual mechanism involving both renal and respiratory regulation, this system underscores the complexity and marvel of biological homeostasis.