Welcome to our comprehensive guide on understanding calcium ion charge. This is where we dive deep into the fascinating world of chemistry and demystify one of the fundamental concepts: the charge of calcium ions. Calcium ions are a cornerstone in various fields, including biology, chemistry, and even materials science. Whether you're a student, a professional, or simply curious about how things work, this guide will arm you with the knowledge to grasp calcium ion charge in an accessible and practical way.
Problem-Solution Opening Addressing User Needs
Many find themselves puzzled by the concept of calcium ion charge, especially if they’re dealing with chemistry for the first time. The confusion often arises from the basics of atomic structure and electron configuration. The problem lies in translating these abstract concepts into something understandable about calcium ions. We’re here to bridge that gap, providing clear, actionable advice that not only explains the calcium ion charge but also empowers you to apply this knowledge in real-world scenarios.
Our guide isn’t just about learning; it’s about understanding. We’ll break down complex ideas into simple, digestible parts, ensuring you come away not only knowing what a calcium ion charge is but also why it matters in everyday life and professional settings.
Quick Reference
Quick Reference
- Immediate action item: Start by understanding that an atom’s charge comes from the difference between protons and electrons. For calcium ions, this difference leads to a specific charge.
- Essential tip: Calcium typically loses two electrons to form a calcium ion, which leads to a 2+ charge. This step-by-step guidance helps you understand electron configuration and ionization.
- Common mistake to avoid: Confusing the charge of an ion with the number of protons in an atom. Remember, calcium ions are formed by losing electrons, not protons.
Understanding Atomic Structure and Electron Configuration
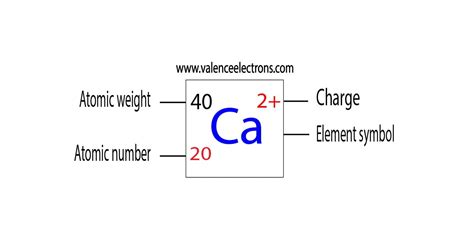
To grasp the calcium ion charge, we need to start with the basics of atomic structure and electron configuration. Calcium, with the atomic number 20, has 20 protons in its nucleus. To understand its charge, we must delve into how electrons are arranged around this nucleus.
An atom is neutral when the number of protons equals the number of electrons. However, calcium ions don’t exist as neutral atoms. Calcium ions are formed when calcium atoms lose electrons, a process known as ionization. Specifically, calcium loses two electrons to achieve stability, leading to a calcium ion with a 2+ charge.
Here's a step-by-step breakdown:
- Neutral calcium atom: Contains 20 protons and 20 electrons, resulting in a neutral charge.
- Ionization process: Calcium loses two electrons, reducing its electron count by 2.
- Resulting charge: With 20 protons still in the nucleus but only 18 electrons orbiting, the atom (now a calcium ion) carries a 2+ charge.
This charge is crucial in various chemical reactions and biological processes, highlighting the importance of understanding calcium ion charge.
Why Does Calcium Lose Two Electrons?
Understanding why calcium loses two electrons to form a 2+ ion involves delving into electron configuration and the concept of energy levels. Calcium’s electron configuration is 1s² 2s² 2p⁶ 3s² 3p⁶ 4s². The outermost electrons, known as valence electrons, are in the 4s orbital. Calcium is stable when it loses these two electrons, achieving a noble gas configuration similar to argon, which has a full outer shell.
This loss of two electrons reduces the repulsive forces within the electron cloud and minimizes the atom’s overall energy, making the calcium ion stable and essential for various biological functions, such as bone mineralization and signal transmission.
The Chemistry Behind Calcium Ion Charge
At the heart of calcium ion charge is the concept of electronegativity and ionic bonds. Calcium ions form by losing two electrons, creating a positive charge that attracts negatively charged ions, such as chloride or phosphate, forming compounds like calcium chloride (CaCl₂) and calcium phosphate (Ca₃(PO₄)₂).
Here’s how it works:
- Electronegativity: Calcium’s electronegativity is lower than many other elements, making it easier for calcium to lose electrons rather than gain them.
- Ionic bonds: The attraction between calcium ion (Ca²⁺) and anions (like Cl⁻) leads to the formation of ionic compounds, where calcium’s 2+ charge is balanced by two chloride ions, each with a 1- charge.
Real-World Applications of Calcium Ion Charge
Understanding the calcium ion charge isn’t just an academic exercise; it has profound implications in real-world scenarios. From the human body’s functioning to industrial applications, calcium ions play a pivotal role.
In biological systems, calcium ions are vital for muscle contraction, blood clotting, and nerve function. The concentration of calcium ions in the blood is tightly regulated to ensure these processes work correctly. In the industrial realm, calcium ions are used in manufacturing processes, such as in the production of cement and glass, where they help form calcium silicates and calcium aluminate.
Practical Tips and Best Practices
Here are some practical tips and best practices for understanding and working with calcium ion charge:
- Visualize electron configuration: Use diagrams or software to visualize how calcium loses two electrons to form its ion.
- Practice with other ions: Apply the concept of ionization to other elements to understand how different ions form.
- Experiment safely: If working in a lab, handle calcium and its compounds with care, following all safety protocols.
Practical FAQ
What is the significance of calcium ion charge in biological systems?
Calcium ions play a crucial role in many biological processes due to their ability to form stable ionic bonds. They are essential for muscle contraction through the regulation of actin and myosin filaments, blood clotting by activating the coagulation cascade, and neurotransmitter release. Calcium ions also help maintain the structural integrity of bones and teeth through the formation of calcium phosphate crystals.
Can you give an example of a common mistake when working with calcium ions?
A common mistake is confusing the charge of an ion with the number of protons in an atom. It’s important to remember that calcium ions are formed by losing electrons, not by changing the number of protons. This is a fundamental concept in understanding why calcium forms a 2+ ion.
Advanced Concepts: The Role of Calcium Ions in Materials Science
For those looking to delve deeper, understanding calcium ions in materials science can open up a world of advanced applications. Calcium ions are pivotal in creating certain types of cement and glass. The chemistry behind these materials involves calcium silicates and aluminates, where calcium ions help form strong, durable structures.
In advanced research, calcium ions are also studied in the context of electrochemical applications, such as in batteries and capacitors, where their ability to move freely within an electrolyte solution can store and release energy efficiently.
This guide has aimed to simplify the concept of calcium ion charge, providing you with a solid understanding of why and how calcium loses electrons to form its 2+ charge. By integrating practical examples and addressing common questions, we hope to have demystified calcium ion charge for you, making it an accessible and valuable piece of knowledge.