To navigate the often confusing waters of molecular polarity, it’s essential to understand the difference between polar and nonpolar molecules, especially when dealing with compounds like CF4 (carbon tetrafluoride). This guide will walk you through identifying molecular polarity with step-by-step guidance and practical examples to ensure that you can confidently determine if CF4 is polar or nonpolar. Let’s dive into the exciting world of molecular polarity, starting with the fundamental problem it presents.
Understanding the Problem: Identifying Molecular Polarity
Determining molecular polarity can be a challenging task for many students and professionals alike. It’s crucial for understanding chemical reactivity, solubility, and physical properties. Misidentifying whether a molecule is polar or nonpolar can lead to incorrect conclusions in various scientific applications. Whether you’re a chemistry student or a professional in a related field, understanding molecular polarity can significantly impact your work. This guide will provide the actionable steps and insights you need to master molecular polarity.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Draw the Lewis structure of the molecule and assess the electronegativity differences between atoms to identify polarity.
- Essential tip with step-by-step guidance: First, sum up the valence electrons, place the least electronegative atom in the center, and arrange more electronegative atoms around it. Use the formula: (3.5 - Electronegativity Difference) * Number of Bonds for assessing bond polarity.
- Common mistake to avoid with solution: Don’t forget to consider the molecular geometry; asymmetrical structures are typically polar while symmetrical ones are nonpolar.
Decoding CF4: Step-by-Step Identification of Polarity
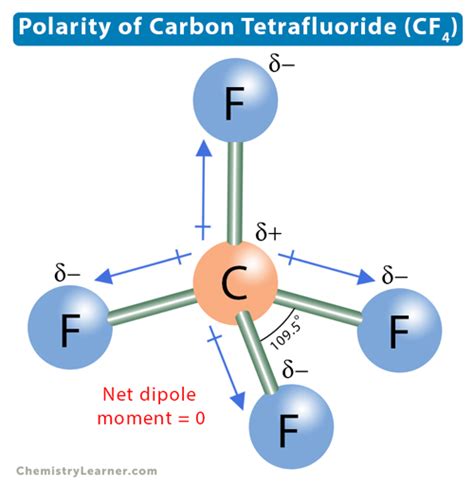
CF4 is composed of one carbon atom and four fluorine atoms. To determine its polarity, we need to break down the steps systematically. Let’s start by looking at the basics.
Step 1: Drawing the Lewis Structure
1. Count Valence Electrons: Carbon has 4, and each fluorine atom has 7. Total = 4 + (4 * 7) = 32 valence electrons. 2. Place Central Atom: Carbon goes in the center surrounded by four fluorine atoms. 3. Form Bonds: Distribute electrons to form four C-F single bonds. Each bond takes two electrons, using 8 out of the 32 electrons. 4. Complete Octets: Fluorines are surrounded by eight electrons (complete octet), and carbon has four lone pairs around it (in reality, it only has two lone pairs since it’s using all 32 electrons).
Step 2: Electronegativity Differences
1. Electronegativity Values: Carbon is around 2.5 and Fluorine around 4.0. 2. Difference: The difference is 1.5, indicating a polar bond since it’s above the threshold where bond polarity starts to matter (usually 0.5).
Step 3: Assess Molecular Geometry
1. Molecular Shape: CF4 has a tetrahedral shape. 2. Symmetry: Even though each C-F bond is polar due to electronegativity differences, the tetrahedral symmetry causes the dipoles to cancel each other out, leading to a nonpolar molecule.
This step-by-step breakdown shows us why CF4 is nonpolar, despite having polar bonds. Now, let’s move on to another practical scenario.
Detailed Steps to Verify Polarity in Any Molecule
When examining any molecule for polarity, follow these comprehensive steps to ensure accurate results:
Step 1: Determine Electronegativity Values
Calculate the difference in electronegativity between the bonded atoms. For example, between carbon (2.5) and fluorine (4.0). An electronegativity difference greater than 0.5 usually indicates a polar bond.
Step 2: Draw the Lewis Structure
1. Start by sketching the skeleton structure where the least electronegative atom (often the central atom) is placed first. 2. Connect the atoms with single bonds and place the remaining electrons around outer atoms to complete their octets. 3. If there are lone pairs on the central atom, note them as they play a crucial role in molecular geometry.
Step 3: Identify the Molecular Geometry
Use the VSEPR (Valence Shell Electron Pair Repulsion) theory to predict the shape of the molecule. This will help you understand how the individual bond dipoles will interact.
Step 4: Assess the Molecular Symmetry
A molecule is polar if it has a net dipole moment; this can be determined by its overall shape. Molecules with symmetrical geometry, such as linear, trigonal planar, or tetrahedral configurations with evenly distributed dipoles, often end up being nonpolar.
Here are practical examples for better understanding:
| Molecule | Shape | Electronegativity Difference | Symmetry | Polarity |
|---|---|---|---|---|
| NH3 | Trigonal pyramidal | 0.94 (N-H) | Asymmetrical | Polar |
| CO2 | Linear | 0.89 (C-O) | Symmetrical (linear cancels out dipoles) | Nonpolar |
This table showcases two different molecules, highlighting the critical factors contributing to their polarity.
Practical FAQ: Addressing Common Questions
What makes a molecule polar or nonpolar?
A molecule is considered polar if it has an uneven distribution of electron density, leading to a dipole moment. This typically occurs due to differences in electronegativity between bonded atoms and an asymmetrical molecular geometry. Conversely, if the molecule’s shape allows the dipoles to cancel out, the molecule is nonpolar.
How can I quickly check if a molecule is polar without a Lewis structure?
Look for molecules with highly electronegative atoms like fluorine, oxygen, or nitrogen bonded to less electronegative atoms. If they have a symmetrical geometry (like tetrahedral or linear), chances are they’re nonpolar. For asymmetrical molecules like trigonal pyramidal or bent shapes, they are likely polar.
Can a molecule with polar bonds still be nonpolar?
Yes, a molecule can have polar bonds but still be nonpolar if its geometry allows the bond dipoles to cancel each other out. This often happens in symmetrical shapes like tetrahedral or linear.
This comprehensive guide is designed to help you confidently determine the polarity of molecules. Whether you are tackling CF4 or any other compound, understanding the principles outlined here will be invaluable. Keep practicing these steps, and you’ll soon master the art of identifying molecular polarity.