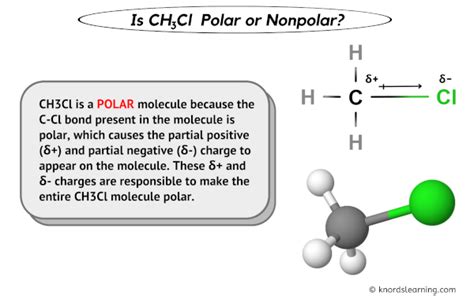
Is CH3Cl Polar or Nonpolar? Unraveling Its Chemistry Secrets
Methyl chloride, also known as CH3Cl, is an organic compound that finds relevance in various industrial applications, from pharmaceuticals to organic synthesis. Understanding its polarity is essential, given that polarity affects the substance’s physical properties and reactivity. Delving into CH3Cl’s molecular geometry and electron distribution will reveal whether this molecule leans towards the polar or nonpolar spectrum. This piece aims to dissect the molecular characteristics and reveal why CH3Cl is categorized as a polar molecule.
Key Insights
- Primary insight with practical relevance: CH3Cl's molecular shape and electronegativity differences make it a polar molecule, which impacts its behavior in chemical reactions and solubility.
- Technical consideration with clear application: The asymmetric distribution of charge around the carbon atom due to the presence of the chlorine atom leads to a net dipole moment, confirming its polar nature.
- Actionable recommendation: For industrial applications, the polarity of CH3Cl should be considered when designing processes that involve its use to optimize efficiency and safety.
Understanding CH3Cl's Molecular Structure
To ascertain the polarity of CH3Cl, one must first comprehend its molecular structure. The molecule comprises a central carbon atom bonded to three hydrogen atoms and one chlorine atom. This arrangement forms a tetrahedral geometry. In this shape, the C-H bonds are uniformly distributed around the carbon atom, with the C-Cl bond extending outward at an angle due to the larger atomic radius of chlorine compared to hydrogen. This asymmetric arrangement of atoms means there is an uneven charge distribution, creating a net dipole moment—a clear indicator of a polar molecule.
Electronegativity and Charge Distribution
Electronegativity plays a pivotal role in determining the polarity of molecules. Chlorine is more electronegative than carbon and hydrogen, resulting in a greater share of electron density around the chlorine atom. The difference in electronegativity between the carbon and chlorine atoms causes a partial positive charge to reside on the carbon atom, while the chlorine atom carries a partial negative charge. The absence of symmetry in this charge distribution confirms that CH3Cl is polar.
The polar nature of CH3Cl affects its interactions with other molecules, influencing its solubility in polar solvents like water, its boiling and melting points, and its reactivity in chemical processes.
Why is the polarity of CH3Cl important in chemical reactions?
Understanding the polarity of CH3Cl is crucial for predicting its behavior in chemical reactions. Polar molecules like CH3Cl are more reactive with other polar substances and can participate in dipole-dipole interactions, hydrogen bonding, and other intermolecular forces, which can significantly affect reaction mechanisms and products.
How does CH3Cl's polarity affect its industrial applications?
In industrial settings, the polarity of CH3Cl is leveraged to enhance its solubility in polar solvents for extraction processes. Its polar nature also makes it useful in synthesis pathways where specific reaction conditions favor polar compounds.
In conclusion, the polarity of CH3Cl is a defining characteristic that stems from its molecular structure and electronegativity differences. This polar nature influences its physical properties and chemical reactivity, making it a significant factor to consider in both theoretical and applied chemistry. By understanding these secrets, we can better utilize CH3Cl in a variety of scientific and industrial contexts.