Plunging into the realm of gas laws, Charles Law stands out as a fundamental principle governing the relationship between volume and temperature in gases. Discovered by the French chemist Jacques Charles in 1787, this principle offers invaluable insights into gas behavior under varying thermal conditions. Understanding Charles Law can greatly benefit professionals in fields such as thermodynamics, chemical engineering, and environmental science, where precise control and prediction of gas volume changes are paramount.
Key Insights
- Charles Law illustrates the direct proportionality between volume and temperature.
- Useful for predicting gas behavior in practical engineering applications.
- Employ Charles Law to design systems ensuring optimal gas handling.
Exploring the Charles Law Equation
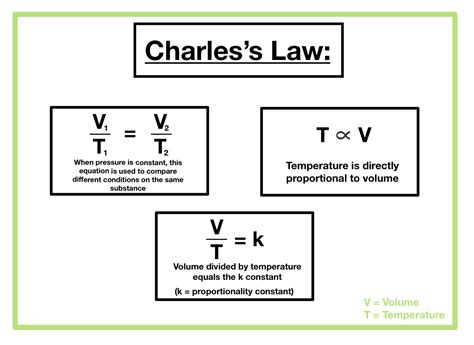
Charles Law, also known as the Law of Ideal Gases, states that the volume of a gas is directly proportional to its absolute temperature when pressure is held constant. The equation for Charles Law is:V / T = constant
Where V represents the volume of the gas, T represents the absolute temperature (in Kelvin), and the constant remains unchanged as long as pressure and the quantity of gas remain constant.
This equation is particularly beneficial in scenarios where thermal changes are predictable, allowing for accurate forecasting of gas volume shifts. For example, in cryogenics, where gases are cooled to extremely low temperatures, Charles Law provides a reliable framework for calculating volume changes, ensuring safety and efficiency in applications such as liquid gas storage and transportation.
Applications of Charles Law in Real World Scenarios
One prominent application of Charles Law is in weather balloon technology. Weather balloons carry instruments into the upper atmosphere to gather data on wind speed, temperature, and pressure. Since the atmosphere’s temperature decreases with altitude, the volume of the gas inside the balloon expands as it ascends. According to Charles Law, if the balloon’s internal pressure remains constant, the gas volume increases as the external temperature drops. This expansion stretches the balloon’s material to its limit, which is crucial for meteorologists in designing these balloons to withstand such expansions safely.In the realm of chemical engineering, Charles Law is used to design reactors where precise gas volume control is essential. By adjusting the temperature, engineers can manipulate the volume of gases inside the reactor, thereby optimizing chemical reactions and improving process efficiency.
How does Charles Law relate to the behavior of gases in everyday life?
Charles Law plays a vital role in everyday activities involving gases. For example, inflating a bicycle tire on a cold day results in less air compared to inflation on a warm day, due to the reduction in volume as the temperature drops. Understanding Charles Law helps in predicting these variations, ensuring the tire has the proper air pressure for safety and comfort.
Can Charles Law be applied at different pressures?
Charles Law specifically pertains to constant pressure conditions. When pressure is not constant, the gas law equations involving Boyle's Law or the Combined Gas Law need to be considered. However, in many practical applications where maintaining consistent pressure is feasible, Charles Law remains highly applicable and useful.
The exploration and application of Charles Law are critical for advancing scientific and engineering disciplines. From weather balloon operations to chemical reactor designs, Charles Law offers foundational knowledge that drives innovation and efficiency across various domains. The precision and predictability provided by this law underscore its significance in both theoretical and applied science.