Are you finding it difficult to grasp the concepts of chemical weathering, especially as they apply to your daily life or specific project needs? Understanding chemical weathering is crucial for fields ranging from geology to environmental science, agriculture, and even urban planning. This guide aims to break down complex processes into easily digestible pieces, giving you real-world examples and actionable tips to enhance your knowledge and practical application.
Why Understanding Chemical Weathering Matters
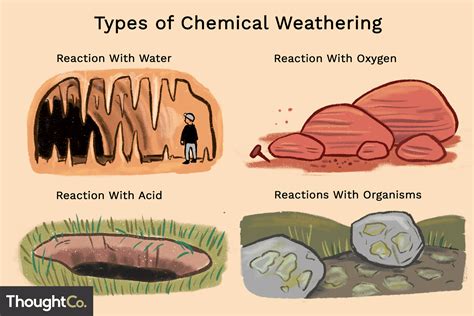
Chemical weathering refers to the process by which rocks and minerals undergo chemical reactions that alter their composition. Unlike physical weathering, which involves breaking down rocks through mechanical forces, chemical weathering changes the material’s internal structure. This process is vital because it contributes to soil formation, affects water quality, influences the carbon cycle, and even plays a role in climate regulation.For instance, when you walk through a forest, the breakdown of rocks due to chemical weathering helps create the nutrient-rich soil that supports plant life. Understanding these processes can help you make informed decisions in areas like land use, sustainable farming, and even urban development. Let’s dive into some actionable steps and examples to understand these concepts better.
Quick Reference
Quick Reference
- Immediate action item: Start by observing the natural weathering of rocks in your area. Notice how different elements like rain, acid, and temperature changes affect the rocks.
- Essential tip: Learn to identify the different types of chemical weathering processes such as hydrolysis, oxidation, and carbonation.
- Common mistake to avoid: Confusing chemical weathering with physical weathering. Remember, chemical weathering involves a change in the chemical composition of the rock, not just its physical breakdown.
The Science of Hydrolysis: Breaking Down Minerals
Hydrolysis is one of the primary forms of chemical weathering where water molecules interact with minerals in a rock to form new minerals and soluble salts. This process is particularly evident in feldspar minerals found in igneous rocks. When feldspar interacts with water, it breaks down into clay minerals and soluble ions.Example:
Imagine you’re studying a rock from a mountain that contains feldspar. Over time, rainwater that carries dissolved CO2 forms carbonic acid, which then reacts with the feldspar to form kaolinite clay and dissolved ions of potassium and other elements. This is how nutrient-rich clay soils develop, essential for agriculture.
Step-by-Step Guidance:
- Identify the minerals in the rock: Use a microscope or X-ray diffraction to determine if your rock contains feldspar.
- Expose the rock to natural weathering: Place samples in areas with high humidity and observe over several months.
- Test for new minerals: After a few months, analyze the samples to see if clay minerals have formed. Use a handheld X-ray fluorescence (XRF) analyzer if available.
- Record findings: Keep detailed notes on changes in mineral composition and environmental conditions like temperature and humidity.
Best Practices:
- Always label and date your samples to track the progression of weathering.
- Ensure you have a controlled environment to compare with naturally weathered samples.
Oxidation: The Role of Oxygen in Weathering
Oxidation occurs when oxygen interacts with minerals in rocks, often leading to rust-like formations in iron-bearing minerals. This process can visibly change the color and texture of the rock.Example:
Consider a granite boulder with abundant quartz and biotite. As the biotite oxidizes, it breaks down into hematite (iron oxide) and quartz, altering the rock’s appearance. This process can also weaken the rock, making it more susceptible to physical weathering.
Step-by-Step Guidance:
- Examine rocks for iron-rich minerals: Look for minerals like biotite, magnetite, and hematite.
- Observe weathering effects: Pay attention to color changes, especially in iron-bearing minerals.
- Measure changes over time: Use a spectrometer to measure the iron content before and after a period of exposure to the elements.
- Document findings: Record any changes in mineral composition and note the environmental conditions.
Best Practices:
- Regularly check your samples to observe how quickly oxidation occurs under different conditions.
- Consider using controlled laboratory settings to isolate the effects of oxidation from other weathering processes.
Carbonation: The Acid Rain Effect
Carbonation is a chemical weathering process that occurs when rainwater, charged with carbon dioxide from the atmosphere, reacts with minerals in the rock. This is especially noticeable in limestone and marble, which contain calcium carbonate.Example:
If you have a limestone rock, rainwater that has absorbed CO2 forms carbonic acid, which reacts with calcium carbonate to form calcium bicarbonate, which is soluble in water. Over time, this leads to the dissolution of limestone, creating caves and sinkholes.
Step-by-Step Guidance:
- Collect rainwater samples: Gather rainwater from different locations to analyze its acidity level.
- Expose limestone samples: Place limestone pieces in these rainwater samples and observe over time.
- Measure dissolution: Use a pH meter to measure the acidity and a scale to measure the weight loss of the limestone.
- Document changes: Keep a log of the rate of dissolution and the pH changes.
Best Practices:
- Ensure you’re consistent with your observations to understand the impact of different acidity levels.
- Compare samples exposed to natural rainwater with those exposed to distilled water to isolate the effect of natural acidity.
Practical FAQ
How does chemical weathering affect agriculture?
Chemical weathering plays a vital role in soil formation, which is crucial for agriculture. As rocks break down, they release essential nutrients like calcium, magnesium, and potassium. These nutrients are absorbed by plant roots, promoting healthy growth. However, excessive chemical weathering can lead to the loss of these nutrients, necessitating the use of fertilizers to maintain soil fertility. To counteract this, farmers can implement soil conservation practices like crop rotation and the use of cover crops, which help maintain the balance and enhance soil health.