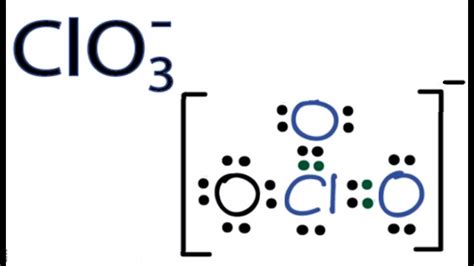Understanding the Lewis structure of ClO3- is fundamental for chemists tackling complex molecular geometry and bonding. This guide is designed to walk you through the intricacies with step-by-step guidance, practical examples, and expert advice to address common challenges you'll face. Let's dive into the molecular world with a problem-solving approach that ensures clarity and comprehension.
Why Understanding ClO3- Lewis Structure Matters
The Lewis structure of ClO3- (chlorate ion) plays a critical role in grasping the basics of molecular geometry, resonance, and ionic bonding. Grasping this structure is key to understanding the behavior of the chlorate ion in various chemical reactions. Misinterpreting this structure could lead to incorrect conclusions about the ion’s reactivity, stability, and its role in redox reactions. This guide will provide actionable advice to ensure you correctly interpret and utilize the ClO3- Lewis structure, enhancing your overall chemical comprehension.
Quick Reference
Quick Reference
- Immediate action item: Count valence electrons for Cl and O atoms.
- Essential tip: Place chlorine in the center, arrange oxygens around it, and connect with single bonds.
- Common mistake to avoid: Forgetting to account for the negative charge on the ion when assigning lone pairs.
Step-by-Step Breakdown of ClO3- Lewis Structure
The first step in understanding the ClO3- Lewis structure involves identifying the valence electrons of chlorine and oxygen atoms. Chlorine, being in Group 17, has 7 valence electrons. Oxygen, in Group 16, has 6. Since ClO3- is an ion, we must consider the additional electron from the negative charge. Here’s how to proceed:
Step 1: Determine Total Valence Electrons
Begin by adding the valence electrons of each atom and considering the ion’s charge.
| Atom | Valence Electrons | Total Contribution |
|---|---|---|
| Chlorine (Cl) | 7 | 7 |
| Oxygen (O) | 6 | 3 × 6 = 18 |
| Extra electron from charge | 1 | 1 |
| Total | 26 |
You start with 26 valence electrons for the ClO3- ion.
Step 2: Structure Layout
Next, position chlorine in the center with three oxygen atoms around it. Use single bonds to connect each oxygen atom to the chlorine. Each single bond accounts for 2 valence electrons, so initially, you use 6 electrons here.
- Structure looks like: Cl - O - O - O
Now, you have used 6 out of your 26 valence electrons.
Step 3: Complete Octets Around Oxygen Atoms
Each oxygen atom should ideally have 8 electrons around it (a complete octet). Since each oxygen already has 2 electrons from the bonds, you need to add 4 more electrons per oxygen atom (1 oxygen atom = 4 electrons, total for 3 oxygen atoms = 12 electrons).
Step 4: Place Remaining Electrons on Chlorine
After providing the extra electrons to the oxygen atoms, you have 14 electrons left. These need to satisfy the remaining electrons required for chlorine to have a complete octet. Chlorine will have 6 electrons from bonds and 4 lone pairs to complete its octet, leaving 6 electrons to be placed as lone pairs.
- Structure with lone pairs on chlorine: Cl (lone pairs) - O - O - O
You now have accounted for all 26 valence electrons.
Step 5: Address the Negative Charge
The negative charge comes from one lone pair on the chlorine atom, fulfilling the ion’s charge.
Practical FAQ
Why does the ClO3- ion have resonance?
The ClO3- ion exhibits resonance due to the delocalization of the negative charge across the three oxygen atoms. This means the actual structure isn’t one static arrangement but rather a hybrid of several structures where the charge is shared equally among the oxygens. Resonance contributes to the stability of the ion, as it spreads out the negative charge, reducing electron-electron repulsion.
How does resonance affect the Cl-O bond lengths in ClO3-?
Resonance leads to a situation where all Cl-O bonds are of equal length, contrary to what you might expect if one bond were a single bond and the other two were double bonds. The electrons involved in bonding are delocalized across all bonds, resulting in a bond order that is between a single and double bond for each. This uniform bond length is a direct consequence of the equal distribution of the electron density due to resonance.
This guide offers a practical, detailed breakdown of the ClO3- Lewis structure, emphasizing practical examples that readers can implement to ensure correct interpretation and application in their studies. Whether you’re tackling an exam or conducting research, understanding the fundamentals presented here will greatly enhance your chemical analysis and problem-solving skills.