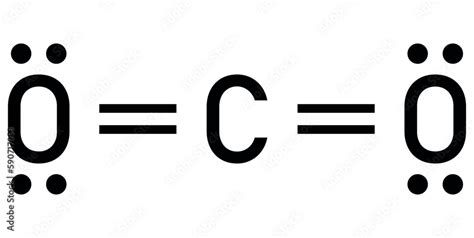Understanding the CO2 Lewis Dot structure is crucial for grasping the fundamental principles of inorganic chemistry. Carbon dioxide, a ubiquitous molecule, provides an excellent case study to explore electron configuration and molecular bonding. This article delves into the CO2 Lewis Dot structure, highlighting practical insights, evidence-based statements, and real examples to elucidate the intricacies of carbon’s electron distribution in this molecule.
Key Insights
- Primary insight with practical relevance: CO2 Lewis Dot structure exemplifies linear molecular geometry and double bond formation.
- Technical consideration with clear application: Understanding the octet rule and electron distribution helps predict chemical behavior.
- Actionable recommendation: Draw accurate Lewis structures to better understand molecular interactions and predict chemical properties.
CO2 Lewis Dot Structure: An Overview
The Lewis Dot structure for CO2 is a pivotal representation of its molecular geometry and bonding. To begin, it’s essential to recall that carbon © is in Group 14, possessing four valence electrons, while each oxygen (O) atom resides in Group 16, carrying six valence electrons. To form CO2, carbon shares electrons with two oxygen atoms, forming two double bonds. Each double bond consists of a pair of shared electrons, which can be represented as pairs of dots around the respective atoms.In a CO2 molecule, carbon assumes a central position, with the two oxygen atoms situated symmetrically on either side, creating a linear structure. This configuration allows for the minimization of electron repulsion, which is consistent with the observed linear geometry.
Octet Rule and Electron Configuration
The octet rule plays a vital role in understanding the CO2 Lewis Dot structure. It states that atoms tend to achieve stability by having eight electrons in their valence shell. In CO2, carbon’s double bonds with each oxygen ensure it fulfills the octet rule. Each oxygen atom, in turn, forms a double bond with carbon and single bonds with lone pairs of electrons, also completing their octet configuration.This adherence to the octet rule is significant, as it provides a foundation for predicting molecular stability and reactivity. For instance, carbon's electron configuration in CO2 is such that it achieves a stable, non-reactive state, which is why CO2 itself is relatively inert under normal conditions.
Molecular Geometry and Bonding
CO2’s molecular geometry is linear, dictated by the arrangement of electron pairs around the central carbon atom. The Lewis Dot structure elucidates this through the representation of two double bonds between carbon and each oxygen atom. The angle between the carbon and oxygen atoms in a CO2 molecule is 180 degrees, a direct consequence of the linear geometry.This linear structure and the absence of lone pairs on the carbon atom indicate no deviations in electron repulsion patterns, resulting in a non-polar molecule. This means that despite two polar bonds (C=O), their arrangement negates any dipole moments, leading to an overall non-polar molecule.
Can the Lewis Dot structure of CO2 change?
No, the Lewis Dot structure of CO2 does not change because it is a stable configuration that satisfies the octet rule for all atoms involved. It represents a state of balance in electron sharing that is energetically favorable.
Why is CO2 considered non-polar?
CO2 is non-polar because it has a linear molecular geometry with a symmetric distribution of charges around the central carbon atom. The two polar C=O bonds are oriented in opposite directions, cancelling each other’s dipole moments and resulting in an overall non-polar molecule.
In conclusion, the CO2 Lewis Dot structure serves as an illustrative example of how to predict molecular geometry, electron sharing, and chemical properties through fundamental principles of inorganic chemistry. The linear arrangement and double bonds exemplify the stability and reactivity trends dictated by the octet rule, providing a robust framework for understanding molecular behavior.