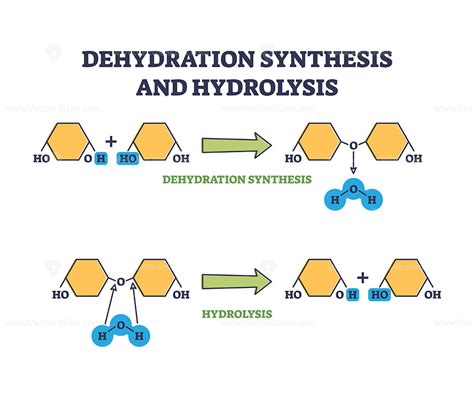
In the realm of biochemistry, dehydration synthesis is a fundamental reaction that plays a crucial role in molecular biology and various biochemical processes. Dehydration synthesis, also known as condensation reactions, facilitates the formation of complex molecules from simpler ones by removing water. This mechanism is essential for forming biopolymers like DNA, RNA, and proteins.
In practical terms, understanding dehydration synthesis is pivotal for scientists working on genetic engineering, pharmaceuticals, and biochemical research. This article will delve into the intricacies of dehydration synthesis, backed by evidence and real-world examples, to provide you with a comprehensive understanding of this important biochemical reaction.
Key Insights
- Primary insight with practical relevance: Dehydration synthesis is a cornerstone process for forming biomolecules, linking simple monomers into complex polymers.
- Technical consideration with clear application: Understanding the energy and catalyst requirements in dehydration synthesis can optimize biological processes and synthetic biology applications.
- Actionable recommendation: Incorporating detailed studies of dehydration synthesis into educational curricula can improve students’ grasp of molecular biology and biochemistry.
Fundamentals of Dehydration Synthesis
Dehydration synthesis involves combining two molecules with the elimination of a water molecule. The chemical equation for this process typically looks like this: A - OH + B - H → A - B + H2O. In this reaction, a hydroxyl group from one molecule and a hydrogen atom from another molecule are removed, forming a bond between the two molecules and water as a byproduct. This reaction is particularly significant in forming macromolecules such as polysaccharides, proteins, and nucleic acids. A practical example is the formation of peptide bonds in proteins, where amino acids join together through dehydration synthesis. Each amino acid in a chain loses a hydroxyl group, while the adjacent amino acid loses a hydrogen atom, resulting in a peptide bond. This process continues to build long chains that fold into functional proteins.Real-World Applications
In biochemistry, dehydration synthesis is indispensable for synthesizing various critical molecules necessary for life. For instance, DNA replication occurs via dehydration synthesis, where nucleotide monomers join to form the DNA double helix. Similarly, in cellular respiration, glucose molecules are broken down through dehydration reactions to produce ATP, the energy currency of cells. An application in synthetic biology is the creation of bioplastics. Researchers are engineering microorganisms to produce biodegradable plastics through dehydration synthesis, which holds promise for reducing plastic waste and addressing environmental concerns.FAQ Section
What distinguishes dehydration synthesis from hydrolysis?
Dehydration synthesis involves the joining of two molecules with the removal of a water molecule, leading to the formation of a larger molecule. In contrast, hydrolysis breaks down a molecule by adding a water molecule, resulting in smaller molecules.
How can we improve the efficiency of dehydration synthesis in laboratory settings?
Optimizing reaction conditions such as temperature, pH, and enzyme concentration can enhance the efficiency of dehydration synthesis. Additionally, utilizing advanced catalysts and exploring biomimetic reactions can further improve reaction rates and yields.
In summary, dehydration synthesis is an essential reaction mechanism in biochemistry with wide-ranging applications from DNA formation to the development of new bioplastics. Through continued research and practical application, we can harness the power of dehydration synthesis to make significant advancements in science and technology.