Understanding the exact density of water, measured in grams per milliliter (g/ml), is fundamental for numerous scientific disciplines and practical applications. While common knowledge often simplifies this value to 1 g/ml, this article dives deeper to elucidate why and how this exact density measurement is crucial. We’ll explore real-world implications, technical considerations, and practical applications, presenting this complex topic in an accessible, authoritative manner.
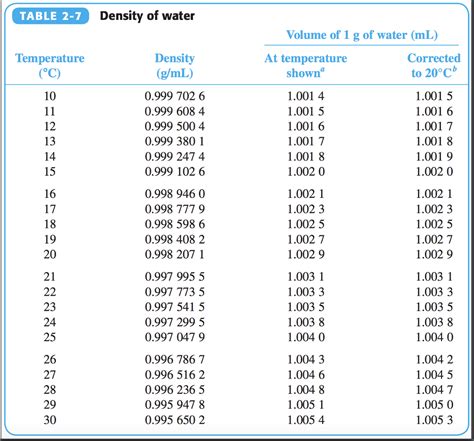
The density of water at 4°C is precisely 0.9998 g/ml, a figure derived from rigorous scientific experimentation. This seemingly small deviation from 1 g/ml carries significant implications in various fields, including hydrology, oceanography, and chemical engineering. To start, the near-perfect density value allows water to serve as an effective medium for buoyancy, influencing the aquatic habitats and ecosystems it supports.
Key Insights
- The density of water at 4°C is precisely 0.9998 g/ml, providing critical implications for buoyancy and ecosystem balance.
- This technical consideration is critical in calculating fluid dynamics in marine environments and designing water treatment systems.
- Understanding the exact density aids in optimizing chemical processes that rely on water as a solvent or reactant.
Importance in Hydrology and Oceanography
In hydrology and oceanography, the precise density of water is fundamental in understanding aquatic ecosystems and oceanic currents. The slight density variation allows water molecules to interact in specific ways, leading to phenomena like thermohaline circulation. This circulation drives global climate patterns, influencing weather systems worldwide. For instance, polar waters, being slightly denser due to lower temperatures, sink and create deep ocean currents that distribute heat across the globe.
Applications in Chemical Engineering
Chemical engineers leverage the exact density of water to design and optimize processes involving water. In industries, precise density values help in formulating accurate equations of state, crucial for designing reactors, separation systems, and for scaling up laboratory findings to industrial processes. For example, when dealing with water-based solutions, engineers must account for the exact density to predict reaction rates, volumes, and material flow, ensuring efficient and safe operation.
How does temperature affect the density of water?
Temperature significantly impacts the density of water, with density peaking at 4°C. Above and below this temperature, water expands or contracts, reducing its density. This thermal expansion and contraction are vital in understanding natural processes such as ice formation and the stratification of lakes and oceans.
Why is the precise density important in water treatment?
Water treatment plants rely on the exact density of water for dosing chemicals accurately. Miscalculating the density can lead to improper chemical concentrations, impacting water purification processes. Understanding the density ensures optimal dosing, resulting in safer drinking water and efficient wastewater treatment.
In conclusion, the exact density of water, 0.9998 g/ml at 4°C, is not merely an academic curiosity; it is a vital factor in a myriad of scientific and practical applications. From influencing marine ecosystems to optimizing chemical processes, precise density measurements underpin many facets of our modern world. By grasping these nuances, professionals can make informed decisions, leading to more effective and sustainable practices across various domains.
Related Terms:
- Density of water kg m3
- Density of salt water