Understanding the subtle yet significant differences between diastereomers and enantiomers is crucial for anyone involved in chemistry, pharmacology, or materials science. These differences dictate how molecules interact with biological systems and the environment, impacting fields as diverse as drug design and the synthesis of advanced materials.
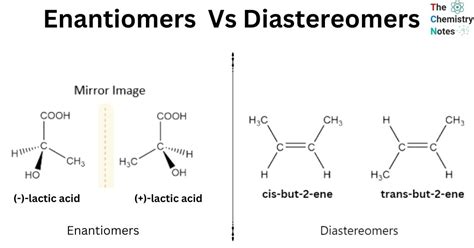
Practical differences between diastereomers and enantiomers are vast and pivotal. While enantiomers are non-superimposable mirror images, diastereomers differ in more than one stereochemical orientation and are not mirror images of each other. Grasping these distinctions is essential for optimizing molecular interactions, ensuring the efficacy of pharmaceuticals, and developing new synthetic pathways.
Key Insights
- Diastereomers are not mirror images and can differ in multiple stereochemical configurations.
- Enantiomers have distinct stereochemistry but share similar chemical properties, differing only in their three-dimensional orientation.
- Practical application: Differentiate between stereoisomers to tailor drug efficacy and selectivity.
Diastereomers: Non-Mirror Image Stereoisomers
Diastereomers are a type of stereoisomer that are not mirror images of each other. They arise when a molecule has two or more chiral centers, resulting in a variation of configurations at one or more of these centers. Unlike enantiomers, diastereomers can interact differently with other molecules, often leading to different physical and chemical properties. A classic example is found in the study of sugars, where glucose and galactose are diastereomers at the fourth carbon position.Enantiomers: Mirror Image Stereoisomers
Enantiomers, in contrast, are non-superimposable mirror images of each other. These stereoisomers share the same connectivity of atoms but differ in the orientation of those atoms in three-dimensional space. One of the most impactful applications of enantiomers is in the pharmaceutical industry, where the biological activity of one enantiomer can be drastically different from its mirror image counterpart. For instance, one enantiomer of a drug may act as an effective pain reliever while the other might cause harmful side effects.The Importance of Diastereomers in Synthesis
In synthetic chemistry, diastereomers often play a critical role in the design of selective reactions. By manipulating the stereochemistry of reactants, chemists can favor the formation of one diastereomer over another, leading to higher yields and purities in the desired product. For example, in the synthesis of complex natural products, controlling diastereoselectivity is crucial for producing compounds with specific biological activity.Enantiomers in Drug Development
In drug development, enantiomers can exhibit dramatically different biological activities, leading to what is known as enantioselectivity. Pharmaceutical companies often discover that only one enantiomer of a drug is effective while the other may be inactive or even detrimental. This phenomenon is highlighted in the case of thalidomide, where one enantiomer was intended to treat morning sickness but the other caused severe birth defects. Thus, enantiomeric purity is vital in ensuring the safety and effectiveness of medications.Can diastereomers be optically active?
Not necessarily. Diastereomers can share the same optical rotation if they have the same chirality at all chiral centers. However, since they are not mirror images, they often exhibit different behaviors in chiral environments.
How do diastereomers differ from constitutional isomers?
Diastereomers differ in the spatial arrangement of atoms but have the same molecular formula and connectivity, while constitutional isomers differ in the connectivity of atoms entirely.
In conclusion, the nuanced differences between diastereomers and enantiomers are fundamental to the fields of chemistry and pharmacology. These distinctions influence how molecules interact and their ultimate applications, emphasizing the importance of understanding stereochemistry in scientific and medical advancements.