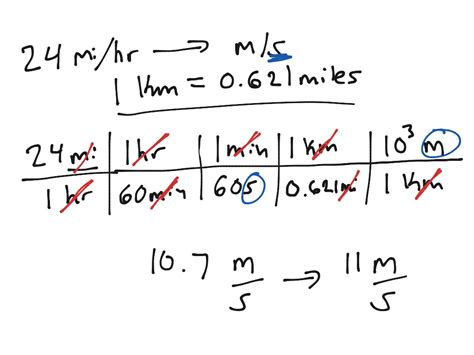If you're tackling chemistry problems and finding it difficult to switch between units, you’re not alone. Dimensional analysis is a powerful technique that can transform your problem-solving experience from frustrating to straightforward. This guide is designed to walk you through the essentials of dimensional analysis in chemistry step-by-step, with actionable advice and practical examples to ensure you can master this crucial skill.
Understanding the Problem: The Pain of Confusing Units
In chemistry, the ability to convert units from one to another is fundamental. This skill is particularly important when dealing with problems involving calculations of rates, concentrations, and conversions between metric and imperial units. The struggle often lies in keeping track of the units and ensuring they cancel out correctly, which is where dimensional analysis comes into play. Mastering dimensional analysis can simplify these conversions, allowing you to solve complex problems more efficiently and accurately.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Always start by writing down all the given units and the units you need to find.
- Essential tip with step-by-step guidance: Use unit conversion factors as fractions that cancel out unwanted units.
- Common mistake to avoid with solution: Avoid multiplying units together; instead, use conversion factors to change one unit into another.
The Basics of Dimensional Analysis
Dimensional analysis, also known as the factor-label method or the unit-factor method, is based on the idea of using conversion factors to change one unit into another. A conversion factor is a ratio that expresses how many of one unit are equal to another unit. Here’s how it works:
You start with what you know and use conversion factors to get to what you need. Each step should include a unit conversion that cancels out the units in the numerator and denominator appropriately.
Let’s consider a simple example: suppose you want to convert meters to centimeters. You know that 1 meter equals 100 centimeters. You can set up your conversion as a fraction:
1 meter / 100 centimeters
You use this fraction in a way that the meters cancel out, leaving you with centimeters.
Detailed How-to: Converting Units Step-by-Step
Here’s a comprehensive step-by-step guide on how to use dimensional analysis to convert units, with practical examples to illustrate each step:
Step 1: Identify Your Starting Unit and Target Unit
Before you start any conversion, clearly identify the units you’re starting with and the units you need to end up with. This clarity is essential to plan your conversion strategy correctly.
Example:
You need to convert 5 kilometers into meters.
Step 2: Find the Correct Conversion Factor
Next, find the conversion factor that relates the starting unit to the target unit. Make sure you know how many of one unit equals the other.
Example:
You know that 1 kilometer equals 1,000 meters.
Step 3: Set Up the Conversion as a Fraction
Write your starting quantity as a fraction, using the conversion factor as needed to convert to the target unit. Always write units next to numbers to ensure they will cancel out properly.
Example:
To convert 5 kilometers to meters:
5 kilometers × (1,000 meters / 1 kilometer) = 5,000 meters
Step 4: Cancel Out Units
In your fraction, multiply and divide, making sure that the units you started with cancel out, leaving you with the units you want.
Example:
5 kilometers × (1,000 meters / 1 kilometer) = 5 × 1,000 meters = 5,000 meters
The “kilometers” unit cancels out, leaving you with meters.
Step 5: Perform the Calculation
Multiply the numbers in your fraction, and make sure the units are correct. This final step will give you the result in the desired unit.
Example:
5 × 1,000 = 5,000
So, 5 kilometers equals 5,000 meters.
Detailed How-to: Handling More Complex Conversions
Sometimes, you’ll need to convert between units that involve more than one step. Here’s how to handle these more complex problems:
Example Problem: Convert 15 liters to milliliters
Step 1: Identify Units
You are converting liters to milliliters.
Step 2: Find Conversion Factor
You know that 1 liter equals 1,000 milliliters.
Step 3: Set Up the Conversion
Write the conversion as a fraction to ensure liters cancel out:
15 liters × (1,000 milliliters / 1 liter)
Step 4: Cancel Units
Ensure that liters cancel out, leaving you with milliliters.
Step 5: Calculate
Multiply the numbers and check the units to get the final result:
15 × 1,000 = 15,000 milliliters
Practical FAQ
What should I do if my units don’t cancel out as expected?
If your units do not cancel as expected, double-check your conversion factors and your set-up. Ensure that you are multiplying and not adding or subtracting units directly. It’s easy to make mistakes, so take your time to verify each step.
How do I handle conversions involving multiple steps?
For multi-step conversions, break the problem down into smaller parts. Convert to a common unit first, and then convert from that common unit to your final target. Always use reliable conversion factors for each step.
In conclusion, mastering dimensional analysis in chemistry is all about practice and precision. By following these steps and understanding the principles behind unit conversions, you can tackle even the most complex problems with confidence and ease. Whether you’re converting between metric units or dealing with more intricate chemistry calculations, dimensional analysis will be your go-to tool for accurate and efficient problem-solving.