Ammonia (NH3) is a fundamental molecule in chemistry that serves as a vital component in many industrial processes and biological systems. Understanding the electron geometry of NH3 is essential for grasping its chemical properties and behavior. This article offers a straightforward guide to the intricacies of NH3 electron geometry, supported by practical insights and real-world examples.
Understanding NH3 Electron Geometry
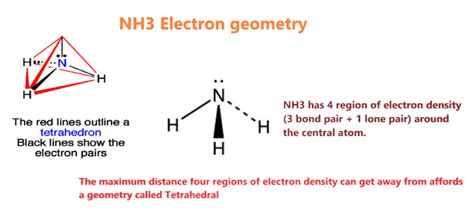
Ammonia’s electron geometry stems from the arrangement of its bonding and lone pair electrons. This knowledge is crucial for predicting its molecular shape, reactivity, and various interactions. In NH3, nitrogen, with its seven valence electrons, forms three sigma bonds with three hydrogen atoms, utilizing three of its electrons, while the remaining four electrons form a lone pair.
The Basics of Electron Geometry
Electron geometry refers to the spatial arrangement of electron pairs around a central atom. In NH3, nitrogen is the central atom. The VSEPR (Valence Shell Electron Pair Repulsion) theory is employed to predict the electron geometry. According to VSEPR, electron pairs will position themselves to minimize repulsion. In NH3, there are four electron groups around nitrogen: three bonding pairs from the nitrogen-hydrogen bonds and one lone pair.
Why Electron Geometry Matters
Understanding the electron geometry of NH3 provides valuable insights into its polarity and chemical interactions. The trigonal pyramidal shape of NH3, resulting from the four electron groups’ spatial arrangement, significantly influences its dipole moment and its role as a base in acid-base reactions.
Key Insights
- Primary insight with practical relevance: Ammonia’s trigonal pyramidal electron geometry is crucial in predicting its polarity and reactivity in various chemical reactions.
- Technical consideration with clear application: The lone pair on nitrogen contributes to NH3's base strength, making it a key player in many biological and industrial processes.
- Actionable recommendation: For optimal understanding, utilize molecular model kits to visually interpret the electron geometry of NH3 and its spatial arrangement.
Applications and Real-World Examples
In agriculture, NH3 is widely used as a fertilizer to enhance soil fertility. Its electron geometry affects its solubility and interaction with soil particles. In chemical synthesis, NH3 acts as a nucleophile due to the lone pair on nitrogen, enabling it to participate in numerous nucleophilic addition and substitution reactions.
Impact on Chemical Behavior
The trigonal pyramidal shape facilitates NH3’s ability to form hydrogen bonds with water molecules, which is why ammonia is highly soluble in water. This solubility is fundamental in various industrial applications, including cleaning agents and in the synthesis of other chemicals.
FAQ Section
What determines the electron geometry of NH3?
The electron geometry of NH3 is determined by the VSEPR theory, which accounts for the four electron groups around the central nitrogen atom, resulting in a trigonal pyramidal shape.
How does NH3’s geometry affect its chemical properties?
NH3’s trigonal pyramidal geometry influences its polarity and ability to act as a base. The lone pair on nitrogen makes it a good nucleophile and enhances its solubility in water.
Understanding the electron geometry of NH3 provides critical insight into its role in various chemical processes and real-world applications. By grasping these concepts, chemists can better predict and manipulate ammonia’s behavior in numerous industrial and biological contexts.