In the realm of chemistry, understanding molecular structures and their configurations is critical. Two fundamental types of isomers often encountered are enantiomers and diastereomers. Their differences are not just academic but have practical implications in various fields including pharmaceuticals, materials science, and biochemistry. This article will demystify these concepts, providing expert perspectives and evidence-based insights.
Understanding Enantiomers
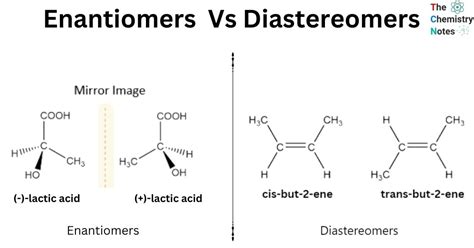
Enantiomers are pairs of molecules that are mirror images of each other, much like left and right hands. They share the same structural formula but differ in their spatial arrangement. This specific spatial configuration is referred to as chirality. Although enantiomers have identical physical properties, they differ in the way they interact with plane-polarized light and other chiral environments. One enantiomer will rotate the light clockwise, while the other rotates it counterclockwise, a property known as optical activity.
Diving into Diastereomers
Diastereomers, on the other hand, are not mirror images of each other. They have different structural formulas and can arise when two or more stereocenters in a molecule have different configurations. Diastereomers differ in multiple stereocenters, leading to a diverse range of physical and chemical properties compared to each other. This distinction is vital when considering their reactivity, solubility, and biological activity.
Key Insights
- Enantiomers are mirror images and exhibit optical activity; diastereomers differ in multiple stereocenters and have distinct physical properties.
- A critical technical consideration is how these isomers behave in chiral environments, affecting areas such as drug efficacy and molecular interactions.
- In practical applications, enantiomer separation is crucial in pharmaceutical synthesis to ensure the desired biological activity.
Implications in Pharmaceuticals
In pharmaceuticals, the distinction between enantiomers and diastereomers is pivotal. The potency and side effects of a drug can often hinge on the chiral nature of the molecule. For instance, one enantiomer of a drug may be therapeutically beneficial, while the other may cause adverse effects. A classic example is thalidomide, where one enantiomer caused severe birth defects, highlighting the need for precise enantiomer separation. Thus, understanding and manipulating chirality can determine the success of drug development and safety.
Applications in Materials Science
In materials science, enantiomers and diastereomers play a role in the development of advanced materials with specific optical properties. For example, certain organic light-emitting diodes (OLEDs) and nonlinear optical materials require precise control over molecular configuration to optimize performance. By studying and synthesizing these isomers, researchers can develop materials with enhanced functionalities that have applications ranging from sensors to communication devices.
Can enantiomers have the same physical properties?
Enantiomers have identical physical properties except for their interaction with plane-polarized light and in chiral environments. They are distinguished by their optical activity.
How are enantiomers and diastereomers separated?
Enantiomers are typically separated using chiral chromatography, while diastereomers can often be separated through conventional chromatographic techniques due to their distinct physical properties.
The nuanced differences between enantiomers and diastereomers underpin much of the molecular behavior observed in complex systems. These insights not only deepen our understanding of chemistry but also drive advancements in medicine and technology. By leveraging this knowledge, scientists can create more effective drugs, develop advanced materials, and further our understanding of molecular interactions. This exploration into enantiomers and diastereomers reveals the intricate beauty and practical applications inherent in molecular structures.