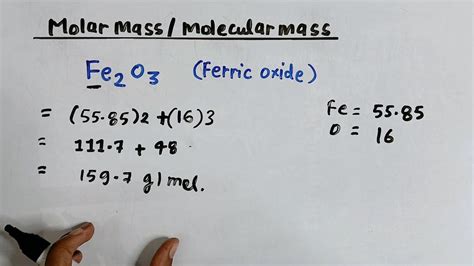The quest for knowledge about the molar mass of iron(III) oxide (Fe2O3) may seem daunting at first, especially for students and professionals navigating the terrain of chemistry. Understanding the concept of molar mass is crucial, as it forms the backbone of stoichiometry, a fundamental aspect of chemistry. This guide aims to demystify Fe2O3 molar mass through step-by-step guidance, actionable advice, and practical examples to address the pain points and queries you might encounter.
Why Knowing Fe2O3 Molar Mass Matters
Understanding the molar mass of Fe2O3 is essential for a variety of reasons. For chemists and students alike, it provides foundational knowledge for calculating chemical reactions, determining the amount of reactants needed, and predicting product quantities. Miscalculating molar mass can lead to errors in laboratory procedures, affecting experimental outcomes. Therefore, grasping this concept ensures accuracy and efficiency in chemical practices.Quick Reference Guide for Fe2O3 Molar Mass
Quick Reference
- Immediate action item: Calculate the molar mass of Fe2O3 by adding the atomic masses of 2 iron (Fe) atoms and 3 oxygen (O) atoms.
- Essential tip: To calculate Fe2O3 molar mass accurately, you need to refer to the periodic table for the atomic masses of Fe and O.
- Common mistake to avoid: One common error is forgetting to multiply the atomic mass of iron by 2 because there are two iron atoms in Fe2O3.
Calculating Fe2O3 Molar Mass: Detailed How-To
Let’s dive into a detailed explanation of calculating the molar mass of Fe2O3. Follow these steps to understand and compute the molar mass accurately.Step-by-Step Calculation
To find the molar mass of Fe2O3, follow these structured steps:
- Identify the elements: Fe2O3 consists of iron (Fe) and oxygen (O). You have two iron atoms and three oxygen atoms.
- Find atomic masses: Look up the atomic masses of iron and oxygen on the periodic table. The atomic mass of iron (Fe) is approximately 55.845 g/mol, and the atomic mass of oxygen (O) is approximately 16.00 g/mol.
- Calculate the molar mass of iron: Since there are two iron atoms in Fe2O3, multiply the atomic mass of iron by 2:
- 2 × 55.845 g/mol = 111.69 g/mol
- Calculate the molar mass of oxygen: There are three oxygen atoms, so multiply the atomic mass of oxygen by 3:
- 3 × 16.00 g/mol = 48.00 g/mol
- Add the molar masses together: Sum the molar masses of iron and oxygen to find the molar mass of Fe2O3:
- 111.69 g/mol + 48.00 g/mol = 159.69 g/mol
Therefore, the molar mass of Fe2O3 is 159.69 g/mol.
Detailed Insights into Fe2O3 Molar Mass
To ensure a deep understanding of Fe2O3 molar mass, let’s delve into additional insights that will strengthen your grasp on the concept.Understanding Atomic Mass Units
The concept of molar mass revolves around atomic mass units (amu). These units represent the mass of atoms relative to the mass of a carbon-12 atom, which is defined as 12 amu. In practical terms, molar mass is expressed in grams per mole (g/mol), which means the mass of one mole of a substance, measured in grams.
Practical Applications of Fe2O3 Molar Mass
Knowing the molar mass of Fe2O3 is not just academic; it has real-world applications. It’s crucial in industries that deal with iron and its compounds, such as steel manufacturing and material science. Understanding molar mass enables the precise formulation of chemical compositions and the execution of chemical reactions.
Accuracy and Precision
Accuracy in calculating molar mass is vital for scientific experiments and industrial processes. Inaccurate molar mass calculations can lead to imbalanced chemical reactions, suboptimal product quality, and even safety hazards. Precision ensures that the exact amounts of reactants are used, leading to desired outcomes.
Real-World Example
Let’s consider a practical scenario in a laboratory setting:
Imagine you need to prepare a solution containing a specific concentration of iron(III) oxide. You must accurately weigh Fe2O3 to achieve this concentration. If your molar mass calculation for Fe2O3 is incorrect, you might use too little or too much Fe2O3, resulting in a non-uniform solution.
Here’s how you’d calculate the required amount:
- Suppose you need 1 mole of Fe2O3 in your solution, and the molar mass of Fe2O3 is 159.69 g/mol. Therefore, you need 159.69 grams of Fe2O3 to prepare the solution.
- Accurately weigh 159.69 grams of Fe2O3 and dissolve it in the appropriate solvent to reach the desired volume of your solution.
This approach ensures precise preparation of chemical solutions, critical for both laboratory experiments and industrial applications.
Practical FAQ on Fe2O3 Molar Mass
Common user question about practical application
What happens if I miscalculate the molar mass of Fe2O3 in a chemical reaction?
Miscalculating the molar mass of Fe2O3 can lead to significant errors in chemical reactions. If you use too much or too little Fe2O3, it can disrupt the stoichiometry of the reaction, resulting in incomplete reactions, side products, and inefficient use of reactants. This can lead to wasted resources and time. To avoid this, always double-check your molar mass calculations and use precise measuring tools.
How can I ensure the accuracy of my molar mass calculations?
To ensure the accuracy of your molar mass calculations for Fe2O3, follow these steps:
- Verify the atomic masses of iron and oxygen from a reliable source like the periodic table.
- Accurately count the number of each type of atom in the chemical formula.
- Multiply the atomic mass of each element by the number of its atoms in the compound.
- Add the results to get the total molar mass.
- Double-check your calculations to avoid any arithmetic mistakes.
Precision in each step ensures the accuracy of your final molar mass calculation.
By following this guide, you can navigate the complexities of Fe2O3 molar mass with confidence. With accurate calculations, practical insights, and real-world applications, you’re well-equipped to handle this fundamental concept in chemistry.