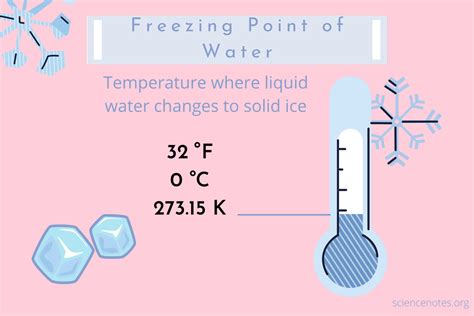
Freezing Point Celsius: Key Temperature Uncovered
Understanding the freezing point of water at 0°C (32°F) is crucial for many everyday activities, from science experiments to cooking and baking. Knowing the exact freezing point can help you troubleshoot issues in the lab, ensure the perfect sorbet in the kitchen, or even prevent pipes from bursting during cold weather. This guide will provide you with the knowledge and practical steps to understand and use this essential temperature, ensuring you can solve any problems that arise.
Problem-Solution Opening Addressing User Needs (250+ words)
Water has an incredible property of transitioning between solid, liquid, and gas states depending on temperature. The freezing point, which is 0°C (32°F), marks the temperature at which water turns into ice. This may sound basic, but the concept is fundamental and frequently encountered in various contexts. Whether you're dealing with scientific experiments that require precise measurements or managing everyday situations like ensuring your home isn't affected by burst pipes during winter, understanding and applying the freezing point of water can be incredibly valuable. Unfortunately, many people either overlook the significance of this temperature or struggle to grasp its practical implications. This guide aims to demystify the freezing point of water and equip you with actionable insights to tackle any related challenges. From basic scientific understanding to real-world applications, this guide will provide step-by-step guidance to ensure you can easily incorporate this key temperature into your everyday life.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: When preparing food, always ensure that the water is at or below 0°C to quickly freeze ingredients.
- Essential tip with step-by-step guidance: To measure the freezing point accurately, use a calibrated thermometer and immerse it in a water sample; watch for a steady temperature of 0°C before concluding it has frozen.
- Common mistake to avoid with solution: Avoid using the refrigerator’s freezer compartment to measure the freezing point as it may not reach the exact 0°C due to variances in internal temperature settings.
Detailed How-To Sections
Understanding and Measuring the Freezing Point of Water
Knowing how to measure the freezing point of water is a fundamental skill for many scientific experiments and practical applications. Here’s a detailed, step-by-step guide on how to measure this key temperature:
Step 1: Gather Materials
You’ll need a calibrated thermometer, a container to hold water, and a source of ice to cool the water. It’s also helpful to have a secondary thermometer for verification purposes.
Step 2: Fill the Container with Water
Fill your container with fresh, clean water. Ensure there’s enough water to immerse the thermometers properly.
Step 3: Prepare the Ice Bath
Fill another container with ice and add cold water. Place the container of water in the ice bath to start cooling the water gradually.
Step 4: Immerse Thermometers
Insert both thermometers into the water. Make sure they are fully submerged but not touching the bottom of the container.
Step 5: Monitor Temperature
Slowly lower the container into the ice bath, and keep the thermometers immersed. Watch the thermometers for any fluctuations. As the temperature drops, it will eventually stabilize at 0°C when water begins to freeze.
Step 6: Verification
Take additional readings from both thermometers. To ensure accuracy, wait for a steady reading at 0°C for a few minutes before concluding that the freezing point has been reached.
Real-World Applications of the Freezing Point
Understanding the freezing point of water is not just for science class. It’s a practical tool in everyday life, from culinary arts to home maintenance.
In Cooking and Baking
For food items like ice creams and sorbets, reaching 0°C ensures a proper freeze. If your kitchen is particularly warm, you might need to pre-chill the ingredients or use ice baths to ensure the freezing point is met.
Preventing Pipe Bursts
During cold weather, it’s vital to ensure water pipes don’t drop below 0°C. If you're going away, make sure to keep your pipes slightly warm or use an insulation wrap to protect them. Leaving any water in a pipe to freeze can cause it to burst.
In Scientific Experiments
For lab experiments, accurately reaching and maintaining the freezing point is crucial for proper sample preparation and results.
Practical FAQ
Why does water expand when it freezes?
Water expands when it freezes because the hydrogen bonds between molecules create a crystalline structure. This structure takes up more space than the liquid state, which is why ice floats on water. Understanding this principle helps explain why water pipes can burst when temperatures drop below 0°C.
What is the safest way to lower the temperature of a water sample to the freezing point?
The safest method to lower the temperature of a water sample to the freezing point is to use an ice bath. Combine ice and cold water in a container, place your water sample in this mixture, and use calibrated thermometers to monitor the temperature. This method allows for controlled and precise cooling.
How can I prevent food from freezing too quickly?
To prevent food from freezing too quickly, you can use a gradual freezing method. Place your food in a container, submerge it in an ice bath as described, but avoid direct ice contact. This will help to moderate the freezing rate, preventing the food from becoming overly firm or icy.
By following this guide, you’ll gain a comprehensive understanding of the freezing point of water, enabling you to effectively use this critical temperature in various practical scenarios.