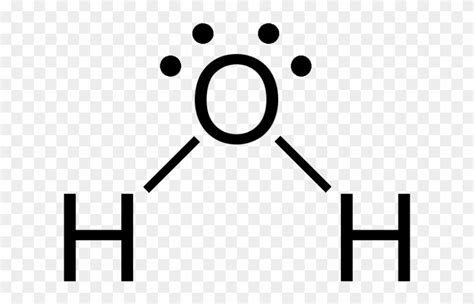
Understanding the Lewis dot structure of water, H2O, can help demystify the chemistry of this ubiquitous molecule. The H2O Lewis dot structure provides insight into how the oxygen and hydrogen atoms form bonds to create a molecule integral to life as we know it. This guide aims to walk you through the journey of discovering the Lewis dot structure of water, highlighting key principles, practical examples, and actionable steps to solidify your understanding.
To many, water is just H2O, but uncovering its chemistry at the molecular level unveils fascinating insights. The Lewis dot structure of H2O allows us to visualize how oxygen shares electrons with two hydrogen atoms, forming a bent molecular geometry. This guide will break down the entire process in a way that's easy to understand, with actionable advice and practical examples to bring these concepts to life.
Introduction: Why Understanding H2O Lewis Dot Structure Matters
The Lewis dot structure is a powerful tool in chemistry that helps us understand the arrangement of atoms in a molecule. For H2O, the Lewis structure reveals how oxygen’s higher electronegativity compared to hydrogen influences bond formation, and how this leads to the water molecule’s distinctive bent shape. Grasping this concept can illuminate many other aspects of chemistry and biochemistry, from molecular polarity to chemical reactions involving water.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Start by counting the valence electrons of oxygen and hydrogen to determine the basic electron-pair setup.
- Essential tip with step-by-step guidance: Follow the octet rule to ensure stability, placing electrons around the central atom first.
- Common mistake to avoid with solution: Don’t forget that oxygen will always be the central atom in the H2O molecule; hydrogen cannot be the central atom as it only needs two electrons.
How to Draw the H2O Lewis Dot Structure
Drawing the Lewis dot structure for H2O involves a few straightforward steps that adhere to fundamental principles of chemistry. Let’s go through each step to ensure clarity and understanding.
Step 1: Determine the Total Number of Valence Electrons
Valence electrons are the outermost electrons that participate in chemical bonding. Oxygen has six valence electrons, and each hydrogen atom has one. For H2O, the total number of valence electrons is:
- 6 (from oxygen) + 2*1 (from two hydrogens) = 8 valence electrons
Step 2: Identify the Central Atom
In most molecules, the least electronegative element is the central atom. Oxygen is more electronegative than hydrogen, so it will be the central atom:
- Central atom: Oxygen (O)
- Surrounding atoms: Two hydrogens (H)
Step 3: Place Electron Pairs Around the Central Atom
Start by placing two electron pairs (four electrons) around the oxygen atom to form bonds with the two hydrogen atoms:
This step gives each hydrogen an electron pair to share, and oxygen has four electrons around it, but we’re not done yet.
Step 4: Complete the Octet Rule for the Central Atom
The octet rule states that atoms like to have eight electrons in their valence shell. Oxygen is nearly there with four electrons, but we need to make it complete with eight by adding lone pairs:
Distribute the remaining four valence electrons as two lone pairs around the oxygen atom:
- Oxygen now has six electrons around it: four from bonding with hydrogen and two lone pairs.
Step 5: Check the Octet Rule for Peripheral Atoms
Each hydrogen atom is satisfied with just two electrons, which is the duet rule for hydrogen (they need only two electrons to achieve stability):
- Each hydrogen has one bond and no lone pairs.
Step 6: Verify the Stability of the Lewis Structure
Re-check your structure to ensure all atoms are stable:
- Oxygen has eight electrons (four bonds and two lone pairs).
- Each hydrogen has two electrons.
With all atoms satisfied, the final Lewis dot structure of H2O looks like this:
| H | O | H | ||
| : | ·· |
Practical Example: Why This Matters in Real-World Chemistry
Consider how this structure explains water’s properties. The bent geometry leads to a polar molecule with a partial negative charge on the oxygen and partial positive charges on the hydrogens. This polarity is why water is an excellent solvent, capable of dissolving a wide variety of substances, from salts to sugars.
FAQ Section
Why does the oxygen atom have two lone pairs in the H2O Lewis structure?
Oxygen has six valence electrons. Four of these are used in bonding with the two hydrogen atoms, leaving two unpaired electrons. These two lone pairs help to achieve the octet rule for oxygen while also contributing to the water molecule’s bent shape and polarity.
What is the significance of the bond angle in the H2O molecule?
The H2O molecule has a bond angle of approximately 104.5 degrees. This angle is less than the typical tetrahedral angle of 109.5 degrees due to the electron repulsion between the lone pairs on the oxygen atom and the bonding pairs with hydrogen. The deviation from the ideal angle illustrates the influence of lone pairs on molecular geometry.
How does the polarity of water affect its properties?
The polarity of water arises from the unequal sharing of electrons between oxygen and hydrogen atoms, leading to a molecular dipole. This results in water being an excellent solvent for ionic and polar substances. Its polarity also contributes to water's high surface tension and boiling/melting points, which are higher than those of similar sized molecules.
By understanding the H2O Lewis dot structure, you can grasp the molecular basis of water’s remarkable properties. This knowledge not only deepens your comprehension of general chemistry but also equips you with insights into more complex chemical behaviors involving water. Follow this guide to build a robust foundation in molecular chemistry and tackle more challenging concepts with confidence.