The process of ion formation is a fundamental concept in chemistry that underlies many phenomena we encounter in everyday life, from the functioning of batteries to the regulation of our body’s electrical signals. Ions are atoms or molecules that possess a net electric charge due to the loss or gain of one or more electrons. This article delves into the mechanics of how ions are formed, offering expert insights and real-world examples to elucidate this crucial aspect of chemistry.
The Basic Mechanism of Ion Formation
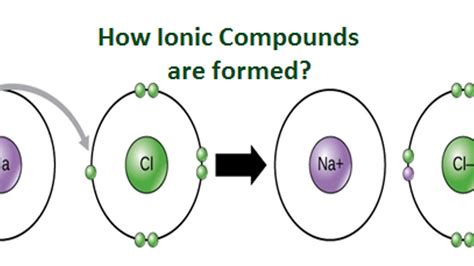
Ion formation occurs when an atom or molecule acquires a net electric charge. This usually happens through the gain or loss of electrons. For instance, when a sodium atom (Na) donates one electron to a chlorine atom (Cl), both become ions. The sodium atom becomes a positively charged sodium ion (Na⁺), while the chlorine atom becomes a negatively charged chloride ion (Cl⁻). The electrostatic attraction between these oppositely charged ions forms an ionic bond, resulting in the compound sodium chloride, commonly known as table salt.
Key insights box:
Key Insights
- Primary insight with practical relevance: Ion formation is central to the formation of ionic compounds, which are prevalent in both natural and industrial applications.
- Technical consideration with clear application: Understanding the specifics of ion formation helps in fields like material science, where ionic compounds are engineered for specific technological uses.
- Actionable recommendation: To deepen comprehension, consider experimenting with simple ionic compound formation in a controlled educational setting.
Ionic Compounds in Everyday Life
The significance of ion formation extends beyond the realm of the laboratory. For instance, in our daily diets, the presence of ions such as sodium and potassium is critical for maintaining fluid balance and nerve function. Moreover, in the industrial sector, ionic compounds are used extensively in the manufacture of glass, ceramics, and even in water purification processes. These applications underscore the critical role ions play in our technological and biological infrastructures.
The Role of Ionization Energy
Ion formation is heavily influenced by ionization energy—the energy required to remove an electron from an atom in its gaseous state. The lower the ionization energy, the easier it is for the atom to become an ion. Conversely, atoms with high ionization energy find it more challenging to lose electrons. For example, noble gases such as helium and neon have exceptionally high ionization energies, making them inert and rarely form ions under normal conditions. Understanding ionization energy is crucial for predicting the likelihood of ion formation in various chemical reactions.
FAQ section:
Can all elements form ions?
Not all elements form ions easily. Elements with low ionization energies, such as alkali metals, readily form positive ions, while elements with high electron affinities, such as halogens, easily form negative ions.
How do ions differ from molecules?
Molecules are neutral entities made up of two or more atoms bonded together. In contrast, ions are charged particles resulting from the loss or gain of electrons, leading to a net positive or negative charge.
In conclusion, the formation of ions is a cornerstone concept in chemistry with widespread implications in both natural and technological contexts. By understanding the basic mechanics of how ions are formed, we gain valuable insight into the behavior of ionic compounds and the essential processes they facilitate in our world.