When it comes to calculating solution concentration, many individuals feel overwhelmed by the complexity and potential for errors. Whether you are a student, a researcher, or someone simply trying to mix solutions for a practical task, understanding the basics and advanced techniques for calculating solution concentrations can save time and reduce frustration. This guide will walk you through everything you need to know, offering practical examples and step-by-step instructions to ensure you master this essential skill.
Understanding Solution Concentration
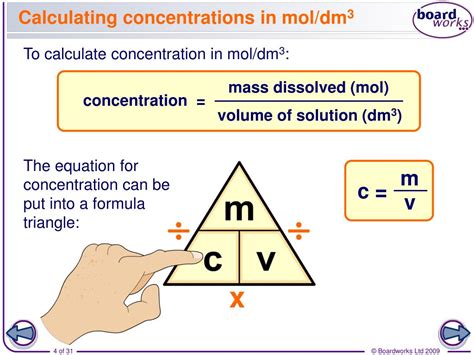
Solution concentration refers to the amount of a solute (the substance being dissolved) in a given quantity of solvent (the substance doing the dissolving). There are several common units of concentration, including molarity (M), molality, and percentage concentration. Understanding these units is crucial for precise calculations.
Why Accuracy Matters
Accuracy in calculating solution concentration is vital in both academic and industrial settings. Inaccurate concentrations can lead to failed experiments, hazardous conditions, and wasted resources. Whether you’re synthesizing chemicals in a lab or preparing solutions for a DIY project, precise concentration calculations ensure consistency and safety.
Quick Reference
Quick Reference
- Immediate action item: Always double-check your calculations and units.
- Essential tip: Use an analytical balance for accurate mass measurements.
- Common mistake to avoid: Confusing molarity (M) with molality (m) and percentage concentration.
Calculating Molarity (M)
Molarity is the most commonly used concentration unit, expressed as moles of solute per liter of solution (M = moles/L). Here’s how to calculate it:
Step-by-Step Calculation
To calculate molarity, follow these steps:
- Determine the number of moles of solute: Use the formula: moles = mass (g) / molar mass (g/mol)
- Measure the volume of the solution: Ensure the volume is in liters (L). You can use a volumetric flask for precise measurements.
- Calculate molarity: Use the formula: Molarity (M) = moles of solute / liters of solution
Let’s work through an example:
- Problem: Calculate the molarity of a solution prepared by dissolving 10 grams of NaCl in enough water to make a 250 mL solution.
- Solution:
- Calculate the moles of NaCl:
- Molar mass of NaCl = 58.44 g/mol
- Moles of NaCl = 10 g / 58.44 g/mol ≈ 0.171 moles
- Convert volume to liters:
- 250 mL = 0.250 L
- Calculate molarity:
- Molarity (M) = 0.171 moles / 0.250 L = 0.684 M
Calculating Molality (m)
Molality is the number of moles of solute per kilogram of solvent (m = moles/kg). It's particularly useful in situations where the solution’s temperature may change, as molality does not depend on volume changes.
Step-by-Step Calculation
To calculate molality, follow these steps:
- Determine the number of moles of solute: Use the formula: moles = mass (g) / molar mass (g/mol)
- Measure the mass of the solvent: Ensure the mass is in kilograms (kg). Use an analytical balance for precision.
- Calculate molality: Use the formula: Molality (m) = moles of solute / kilograms of solvent
Let’s work through an example:
- Problem: Calculate the molality of a solution prepared by dissolving 15 grams of urea in 200 grams of water.
- Solution:
- Calculate the moles of urea:
- Molar mass of urea = 60.06 g/mol
- Moles of urea = 15 g / 60.06 g/mol ≈ 0.250 moles
- Convert the mass of water to kilograms:
- 200 grams = 0.2 kg
- Calculate molality:
- Molality (m) = 0.250 moles / 0.2 kg = 1.25 m
Calculating Percentage Concentration
Percentage concentration can be either mass/mass (%), mass/volume (%), or volume/volume (%). These units are straightforward but can vary significantly in how they are used.
Step-by-Step Calculation
To calculate mass/mass percentage concentration, use the formula:
Mass/Mass (%) = (mass of solute / mass of solution) x 100
To calculate mass/volume percentage concentration, use the formula:
Mass/Volume (%) = (mass of solute / volume of solution) x 100
To calculate volume/volume percentage concentration, use the formula:
Volume/Volume (%) = (volume of solute / volume of solution) x 100
Let’s work through an example:
- Problem: Calculate the mass/mass percentage concentration of a solution prepared by dissolving 20 grams of sodium chloride in 150 grams of water.
- Solution:
- Calculate mass/mass percentage:
- Mass/Mass (%) = (20 g / (20 g + 150 g)) x 100 = (20 g / 170 g) x 100 ≈ 11.76%
Practical FAQ
What is the difference between molarity and molality?
Molarity (M) measures the moles of solute per liter of solution, making it dependent on temperature since volume can change with temperature. Molality (m), on the other hand, measures the moles of solute per kilogram of solvent, which remains constant regardless of temperature changes. Molality is more stable and useful for solutions that undergo significant temperature changes.
How do I convert between different units of concentration?
Conversions between units require knowledge of the given and desired units. For instance, to convert from molarity to molality:
- Calculate moles of solute: moles = molarity x volume (in liters)
- Find the mass of the solvent: Use a balance to get an accurate measurement
- Calculate molality: molality = moles / mass of solvent (in kg)
For conversions involving mass/volume percentage concentration, ensure you have accurate mass and volume measurements.