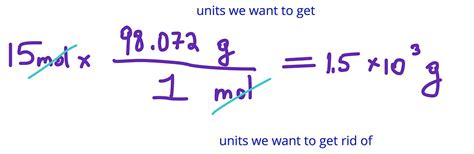Navigating the world of chemistry often involves converting between grams and moles. This fundamental yet complex task is pivotal for balancing chemical equations, understanding molecular quantities, and calculating reaction yields. While it may seem intimidating at first, breaking down this conversion into simple, digestible steps can transform it from a daunting challenge into an accessible process. This guide is designed to provide you with clear, actionable advice and practical solutions for converting grams to moles effortlessly, addressing the user pain points and guiding you through each step with an easy-to-follow approach.
Understanding the Basics: Why Grams to Moles?
The conversion from grams to moles lies at the heart of stoichiometry—the branch of chemistry that deals with the quantitative relationships of reactants and products in chemical reactions. Knowing how to convert grams to moles enables you to determine the amount of a substance in terms of moles, which in turn helps in calculating the amount of other substances involved in a chemical reaction.
The key to this conversion lies in the concept of molar mass, a fundamental property of any substance. The molar mass, typically expressed in grams per mole (g/mol), is the mass of one mole of a given substance. By using the molar mass as a conversion factor, you can easily switch between grams and moles, simplifying complex chemical calculations.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Start with determining the molar mass of the substance you’re working with.
- Essential tip with step-by-step guidance: Use the formula Number of moles = Grams / Molar Mass to convert grams to moles.
- Common mistake to avoid with solution: Always ensure your molar mass is correct, especially when dealing with compounds that contain more than one element.
Detailed How-To: Converting Grams to Moles
Let’s dive into the process of converting grams to moles. Follow these steps to ensure accuracy and ease of understanding.
Step 1: Determine the Molar Mass
The first step is to calculate the molar mass of the substance in question. The molar mass is the sum of the atomic masses of all the atoms in a molecule. This value is often found in the periodic table of elements.
For a single element: If you’re dealing with a simple substance like sodium (Na), the molar mass is simply the atomic mass listed below the element symbol on the periodic table, usually in g/mol.
For compounds: For more complex substances like water (H2O), you need to add up the molar masses of all the atoms in the formula. For water, this would be (2 × 1.008 g/mol for hydrogen) + (1 × 15.999 g/mol for oxygen) = 18.015 g/mol.
Step 2: Use the Conversion Formula
Now that you have the molar mass, you can convert grams to moles using the following formula:
Number of moles = Grams / Molar Mass
Here’s a practical example: suppose you have 25 grams of sodium (Na) and want to find out how many moles this represents. First, look up the molar mass of sodium on the periodic table, which is 22.99 g/mol. Then, apply the formula:
Number of moles = 25 grams / 22.99 g/mol = 1.088 moles
Step 3: Verify Your Calculation
Accuracy is crucial in chemistry. Always double-check your calculations, especially when dealing with decimal values. Utilize scientific calculators or online conversion tools to ensure precision.
Advanced Tips and Tricks
For more advanced conversions, particularly with compounds, consider these tips:
- Practice: Regular practice with different substances helps in quickly grasping the process.
- Dimensional Analysis: Treat your conversion like a math problem, always ensuring that your units cancel out correctly.
- Common Compounds: Familiarize yourself with the molar masses of common compounds to speed up your calculations.
Practical FAQ
What if I don’t have the molar mass?
If the molar mass is not readily available, you can calculate it based on the chemical formula. For instance, for water (H2O), you add the molar mass of two hydrogen atoms (2 x 1.008 g/mol) and one oxygen atom (1 x 15.999 g/mol), resulting in 18.015 g/mol. Alternatively, use a reliable chemical reference or online database for the molar mass of a substance.
Can this process vary for different elements?
Yes, the process remains fundamentally the same regardless of the element or compound. The key difference lies in the specific molar mass of each substance. Always ensure you are using the correct molar mass for accurate conversions.
Why is it important to convert grams to moles?
Converting grams to moles is crucial for several reasons. It helps in determining the amount of reactants needed for a chemical reaction, understanding the quantitative relationships between substances in a reaction, and calculating reaction yields. This conversion forms the backbone of stoichiometric calculations in chemistry.
By following these steps and tips, converting grams to moles will no longer be a challenge. Armed with this knowledge, you’ll be well-equipped to tackle stoichiometric problems with confidence, understanding both the theoretical and practical aspects of chemical conversions.