Is H3PO4 Truly a Strong Acid? Understanding Phosphoric Acid's Role in Chemistry
When delving into the realm of acids, the nomenclature and categorization play a pivotal role in comprehending their behavior in various chemical reactions. Among these, H3PO4, or phosphoric acid, often stirs debate regarding its classification as a strong acid. This guide aims to demystify H3PO4, focusing on its properties, behavior in reactions, and real-world applications, thereby addressing user pain points about its true nature and practical use.
Problem-Solution Opening Addressing User Needs (250+ words)
One of the common dilemmas in chemistry involves discerning whether a substance like H3PO4 fits the category of strong acids. Often, the classification of acids can be confusing, especially when we encounter polyprotic acids such as H3PO4. A clear understanding of phosphoric acid's acidic strength is not only crucial for theoretical knowledge but also for practical applications in fields like food science, industrial processes, and pharmaceuticals.
Many find themselves puzzled about the strength of H3PO4, its dissociation in water, and its behavior in various chemical environments. Misclassification can lead to errors in experimental setups, formulation processes, and even in day-to-day practical tasks. This guide provides a comprehensive, step-by-step analysis of H3PO4, dispelling myths, and presenting actionable insights to help you navigate through its properties and applications confidently.
Quick Reference
Quick Reference
- Immediate action item: Check the pH of a phosphoric acid solution to understand its dissociation.
- Essential tip: Phosphoric acid dissociates in three stages, with the first stage showing stronger acidic behavior.
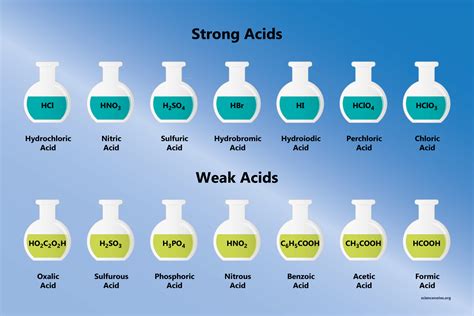
- Common mistake to avoid: Confusing H3PO4 with strong acids like HCl or H2SO4; it's a weak polyprotic acid.
Understanding Phosphoric Acid's Dissociation
Phosphoric acid (H3PO4) is not like your standard monoprotic strong acids. It's a triprotic acid, meaning it can donate three protons in three successive stages of dissociation:
- H3PO4(aq) ⇌ H+(aq) + H2PO4−(aq)
- H2PO4−(aq) ⇌ H+(aq) + HPO42−(aq)
- HPO42−(aq) ⇌ H+(aq) + PO43−(aq)
In each step, the acidic strength diminishes, with the first stage exhibiting the strongest acidic behavior. Unlike strong acids like hydrochloric acid (HCl) or sulfuric acid (H2SO4), phosphoric acid does not fully dissociate in water, classifying it as a weak acid.
How to Measure Phosphoric Acid’s Strength
To understand H3PO4's acidic strength, it’s crucial to explore its dissociation constants, known as Ka values, which quantify the extent of dissociation in each step. The Ka values for phosphoric acid's three dissociation steps are:
- Ka1 ≈ 7.2 x 10⁻³
- Ka2 ≈ 6.3 x 10⁻⁸
- Ka3 ≈ 4.5 x 10⁻¹3
These values underscore phosphoric acid's nature as a weak polyprotic acid, where the first dissociation step has a Ka value within the range of typical strong acids, but the subsequent steps drop significantly.
Here's how to measure its strength practically:
1. Preparation: Prepare a known concentration of phosphoric acid solution.
2. pH Measurement: Utilize a pH meter or pH strips to measure the pH of the solution.
3. Calculate Ka: Using the pH, you can derive the concentration of H+ ions and calculate the dissociation constants for each stage.
For example, if you measure a pH of 2 for a 0.1 M phosphoric acid solution, you can calculate Ka1 using the formula:
Ka1 = [H+][H2PO4−] / [H3PO4]
This step-by-step approach helps grasp how phosphoric acid behaves in solution, reinforcing its weak acidic character.
Practical Applications of Phosphoric Acid
Understanding phosphoric acid's acidic properties opens up a world of practical applications. Here’s how you can leverage this knowledge:
- Food Industry: Phosphoric acid is widely used as a preservative and flavor enhancer in beverages like soda. Its weak acidic nature ensures safe preservation while providing the desired tangy flavor.
- Industrial Uses: In industries, phosphoric acid plays a crucial role in the production of fertilizers, detergents, and various chemical processes. Its controlled acidity aids in optimizing reaction conditions.
- Pharmaceuticals: In drug formulation, phosphoric acid serves as an acidulant and stabilizer, ensuring the pH levels remain within therapeutic ranges.
Practical FAQ Section
Why is H3PO4 not classified as a strong acid?
H3PO4 is classified as a weak polyprotic acid because it only partially dissociates in water. While the first dissociation step has a Ka value similar to strong acids, the subsequent steps show much lower dissociation constants, reflecting its overall weak acidic strength. This partial dissociation is due to the stabilization of the conjugate base at each step, which reduces the acid's strength progressively.
How does the strength of phosphoric acid affect its industrial applications?
The controlled, moderate acidity of phosphoric acid makes it ideal for industrial applications where precise pH control is essential. For example, in detergent manufacturing, the acid’s moderate strength helps maintain optimal pH levels for cleaning efficacy without causing damage to materials. Similarly, in fertilizer production, phosphoric acid’s partial dissociation ensures a steady release of nutrients over time, balancing the needs of plants without over-acidifying the soil.
Best Practices and Tips for Handling Phosphoric Acid
Here are some best practices and tips to keep in mind when working with phosphoric acid:
- Safety First: Always wear appropriate protective gear, including gloves and goggles, when handling phosphoric acid to avoid skin and eye irritation.
- Storage: Store phosphoric acid in a cool, dry place, away from incompatible substances like bases and flammable materials to prevent hazardous reactions.
- Dilution: When preparing solutions, dilute phosphoric acid with water slowly, starting from a concentrated solution, to prevent exothermic reactions that could cause splashing.
Following these guidelines ensures safe and effective use of phosphoric acid in various applications, from lab experiments to industrial processes.
This guide has unveiled the nuances of H3PO4, transforming confusion into clarity and equipping you with the knowledge to appreciate its role in chemistry and its myriad practical applications. With detailed explanations and actionable steps, you can now confidently navigate through the complexities of phosphoric acid’s behavior and leverage its unique properties for your specific needs.