Welcome to our comprehensive guide on heat dynamics, focusing on a fundamental question that's central to cooking, science, and everyday life: Is boiling adding or removing heat? Understanding the dynamics of heat in boiling can transform your cooking, improve your scientific knowledge, and even solve everyday problems at home. This guide is designed to walk you through the principles of heat dynamics during boiling, offering practical insights, actionable advice, and troubleshooting tips to make your understanding clear and applicable. Let's dive right into uncovering the mysteries of boiling and its heat dynamics.
The Core Problem: Misunderstanding Heat Dynamics in Boiling
Many people are puzzled by the heat dynamics of boiling. If you’ve ever wondered why a pot of water boils and then stops bubbling while it continues to heat, or why the temperature remains constant during boiling, you’re not alone. This guide aims to clarify these concepts by exploring the role of heat during boiling. We will break down the process, addressing common misconceptions, and providing real-world examples and solutions. Understanding whether boiling adds or removes heat is crucial for cooking, scientific experiments, and even daily household tasks.
Quick Reference
Quick Reference
- Immediate action item: Start by placing your pot on a stove, heating it, and observe how the water begins to boil. This is where we start understanding heat dynamics in boiling.
- Essential tip: During boiling, the temperature of the water remains constant at the boiling point, regardless of the amount of heat applied, until all the liquid has turned to steam.
- Common mistake to avoid: Assuming that boiling water is always heating up when you see bubbles. It’s critical to understand that while bubbles indicate boiling, the temperature does not increase above the boiling point.
Detailed Understanding of Heat Dynamics During Boiling
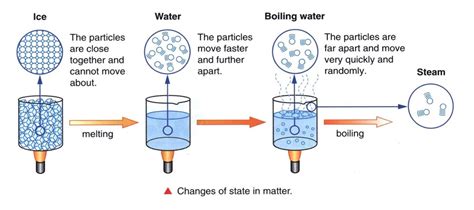
Boiling occurs when water reaches its boiling point, which is 100 degrees Celsius (212 degrees Fahrenheit) at standard atmospheric pressure. During boiling, the water absorbs heat energy, but its temperature does not increase above the boiling point until all the liquid has turned to steam. This section delves into the intricacies of heat dynamics during boiling, breaking down the process step-by-step.
The energy added during boiling is used to break the intermolecular forces holding the water molecules together in a liquid state, allowing them to transition into the gaseous phase. This process requires a specific amount of energy per unit of mass, known as the latent heat of vaporization. For water, this value is approximately 2,260 kJ/kg. While the water is boiling, the energy you're adding goes into changing the state of the water rather than increasing its temperature.
Let's say you're boiling water in a pot on a stove. As you turn up the heat, the water begins to boil, and you'll notice bubbles forming and rising to the surface. Despite the bubbling, the temperature of the water remains at 100°C (212°F). This is because the heat you're adding is being used to turn the water into steam, not to increase its temperature. The energy you add is absorbed as latent heat, which facilitates the phase change from liquid to gas.
This is why, even when the bubbles are vigorous, the water's temperature doesn't rise. It's a crucial concept in understanding boiling dynamics and applying this knowledge in various scenarios, from cooking to scientific experiments.
Practical Examples of Heat Dynamics in Boiling
Understanding heat dynamics in boiling can make a significant difference in your cooking methods and even in scientific experiments. Let’s explore some practical examples to illustrate this further.
Consider boiling pasta. Traditional boiling involves heating a large pot of water until it reaches its boiling point, then adding the pasta. As the pasta cooks, the water absorbs heat, and if you notice, the bubbling slows down as the pasta absorbs heat and the water cools slightly. This is a natural part of the heat dynamics of boiling. To keep the water boiling and cook your pasta evenly, you might need to adjust the heat. This example shows how heat dynamics play out in a familiar cooking process.
In scientific experiments, understanding that boiling water doesn't increase in temperature during the phase change can be vital. For instance, when calibrating thermometers or conducting thermal experiments, knowing that the boiling point remains constant despite heat application can help in accurately measuring temperatures and understanding heat transfer processes.
Practical FAQ: Boiling Water and Heat Dynamics
Why doesn’t the temperature of boiling water increase above the boiling point?
During boiling, the energy you add goes into breaking the bonds between water molecules, changing the state from liquid to gas. This process requires a specific amount of energy, known as the latent heat of vaporization. While the water is boiling, the energy input is used for this phase change rather than increasing the temperature. Hence, the temperature remains constant at the boiling point until all the liquid has turned to steam.
This FAQ section addresses a common question about the practical application of heat dynamics during boiling. By understanding why the temperature remains constant, you can better manage cooking times and temperatures, or conduct more precise scientific experiments.
In conclusion, understanding whether boiling adds or removes heat revolves around recognizing the concept of latent heat and how it facilitates the phase change from liquid to gas without increasing the temperature. This knowledge is not only fascinating but also incredibly practical, offering valuable insights into cooking, science, and everyday life. By following this guide, you've taken a significant step toward mastering the dynamics of boiling and applying this understanding to various aspects of your life.
Remember, the next time you watch water boil, you’re witnessing a complex interplay of heat dynamics at work, and now you have the knowledge to understand it fully. Whether you’re cooking a meal, conducting an experiment, or just curious about the science behind boiling water, you’re equipped with the information to delve deeper into the fascinating world of heat dynamics.