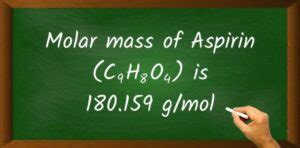Understanding the molar mass of aspirin is crucial for anyone engaged in pharmaceutical chemistry, biochemistry, or related fields. Accurate calculations of molar mass are fundamental to determining dosages, preparing solutions, and understanding the compound’s behavior in various chemical reactions. In this article, we will delve into the detailed process for calculating the molar mass of aspirin and its practical implications.
Calculating the Molar Mass of Aspirin
The molar mass of a compound is a cornerstone of chemistry, defined as the mass of one mole of that substance. Aspirin, chemically known as acetylsalicylic acid, has the molecular formula C9H8O4. To determine its molar mass, we need to sum the atomic masses of all the atoms in the molecule. Using standard atomic masses, we calculate as follows:
- Carbon ©: 12.01 g/mol × 9 = 108.09 g/mol
- Hydrogen (H): 1.008 g/mol × 8 = 8.064 g/mol
- Oxygen (O): 16.00 g/mol × 4 = 64.00 g/mol
Adding these together gives us the molar mass of aspirin:
108.09 g/mol © + 8.064 g/mol (H) + 64.00 g/mol (O) = 180.154 g/mol
Why Molar Mass Matters in Practical Applications
Knowing the molar mass of aspirin enables chemists and pharmacists to prepare precise dosages, ensuring both efficacy and safety. For instance, when creating a buffer solution for a lab experiment, precise molar quantities are vital to achieve the desired pH and concentration levels. The molar mass provides the basis for these calculations, ensuring accurate replication across various experimental setups.
Key Insights
- Calculating the molar mass of aspirin involves summing the atomic masses of its constituent elements.
- This calculation is fundamental for preparing accurate pharmaceutical formulations and conducting precise laboratory experiments.
- Utilizing the molar mass ensures the correct dosage and concentration in both medical and scientific applications.
Real-World Example: Synthesis and Application
Aspirin’s molar mass plays a significant role in its synthesis and application. When synthesizing aspirin, the reactants are combined in stoichiometric ratios based on their molar masses. Typically, salicylic acid and acetic anhydride are used in a reaction where:
C7H6O3 + C4H6O3 → C9H8O4 + C2H4O2
Here, the molar mass calculation helps ensure that precise ratios of reactants are used to yield the desired amount of aspirin. Moreover, when formulating medications, understanding the molar mass allows for correct tablet dosage calculations to avoid therapeutic inefficacy or toxicity.
Advanced Considerations in Molar Mass Usage
In advanced pharmaceutical and biochemical contexts, molar mass calculations go beyond simple dosage formulations. They enable the assessment of molecular behavior in different solvents, stability studies over time, and the interaction with biological molecules such as enzymes. For instance, determining the solubility of aspirin in water involves knowing its molar mass, alongside the knowledge of how it interacts with water molecules.
Understanding aspirin’s molar mass also aids in kinetic studies, where the rate of reaction and the effectiveness of aspirin as an anti-inflammatory agent can be measured. The molar mass is intrinsic to these studies, as it informs the amount of the drug interacting with cellular targets.
How does molar mass relate to aspirin’s efficacy?
The molar mass of aspirin is crucial in determining the precise dosage required to achieve therapeutic efficacy while avoiding adverse effects. Accurate dosing ensures that the correct concentration of the active ingredient is delivered to the patient.
Can molar mass calculations vary due to impurities?
While molar mass calculations are theoretically constant, impurities in aspirin samples can affect the practical mass measurements. Ensuring high purity in aspirin preparations is necessary to maintain accurate dosing and therapeutic outcomes.
This detailed exploration into the molar mass of aspirin underscores its importance in both theoretical and practical applications. From synthesis to dosage, the precise calculation of molar mass ensures the reliability and efficacy of aspirin as a critical pharmaceutical compound.