The molar mass of propane, a commonly used hydrocarbon, is an essential concept for chemists and students alike. Understanding its calculation provides a robust foundation for more advanced topics in chemistry and related fields. Propane’s formula, C₃H₈, might appear straightforward, but the precision in its molar mass calculation is critical for applications in chemical reactions, stoichiometry, and thermodynamics.
Key insights
Key Insights
- Propane’s molar mass calculation is fundamental for accurate chemical analysis.
- Precision in calculation is necessary to avoid errors in chemical reactions and formulations.
- Using a precise molar mass aids in determining the quantities of reactants and products in chemical equations.
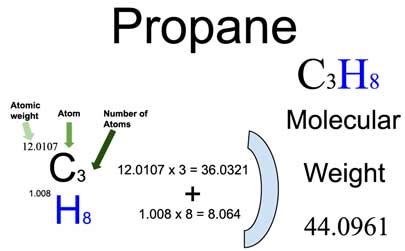
The molar mass of propane is determined by summing the atomic masses of its constituent elements. The molecular formula for propane is C₃H₈, meaning it contains three carbon atoms and eight hydrogen atoms. Carbon’s atomic mass is approximately 12.01 g/mol, and hydrogen’s is roughly 1.008 g/mol. To calculate the molar mass of propane, we multiply the atomic mass of each element by the number of its respective atoms in the molecule and then sum these values.
Detailed Calculation
To start, we calculate the contribution of carbon atoms. There are three carbon atoms, and each has an atomic mass of 12.01 g/mol. Therefore, the total mass from carbon atoms is 3 × 12.01 g/mol = 36.03 g/mol. Next, we determine the contribution from hydrogen atoms. Each hydrogen atom has an atomic mass of 1.008 g/mol, and since there are eight hydrogen atoms, the total mass from hydrogen is 8 × 1.008 g/mol = 8.064 g/mol. Adding these together, the molar mass of propane is 36.03 g/mol + 8.064 g/mol = 44.094 g/mol.Applications and Relevance
This precise calculation of propane’s molar mass is pivotal in various fields. In industrial chemistry, accurate molar mass calculations ensure that the right amount of propane is used in processes such as fuel production and storage. In the laboratory, precise molar mass determination is critical for the accurate formulation of solutions and the execution of chemical reactions. This is particularly important when determining stoichiometric coefficients in balanced chemical equations. Furthermore, accurate molar mass calculations enhance the precision of calorimetry experiments, where the quantity of a substance directly correlates to energy measurements.Practical Example
Consider a scenario where a chemist needs to prepare a solution containing a specific number of moles of propane for an experiment. Using the molar mass of 44.094 g/mol, the chemist can precisely determine the required mass of propane needed for their solution. For example, if 0.5 moles of propane are needed, the mass calculation would be 0.5 moles × 44.094 g/mol = 22.047 grams. Such precision ensures the experiment’s success and avoids the potential hazards of incorrect measurements.Why is the molar mass of propane important in industrial applications?
The molar mass of propane is crucial in industrial applications for the precise calculation of fuel quantities and energy content. Accurate determination ensures efficient fuel usage, cost-effectiveness, and safety in operations like gas storage and transportation.
How does an error in calculating the molar mass affect chemical reactions?
An error in calculating the molar mass can lead to incorrect proportions of reactants and products, affecting the yield and safety of chemical reactions. Inaccurate molar mass measurements can result in inefficiencies, unwanted by-products, and even hazardous situations if safety protocols are compromised.
In conclusion, understanding the precise molar mass of propane underscores the importance of meticulous scientific calculations. From laboratory settings to industrial applications, accurate molar mass determination ensures precision and safety in chemical processes.