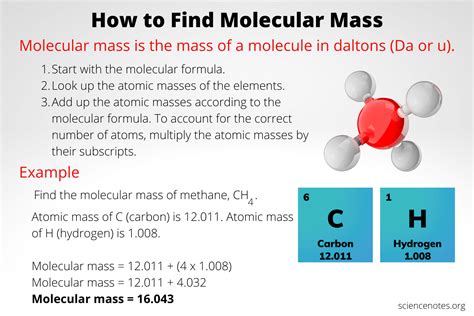
Understanding the molecular mass of a compound is crucial in chemistry as it forms the foundation for various calculations related to chemical reactions, stoichiometry, and even biochemical pathways. In this guide, we’ll demystify the molecular mass of carbon-based compounds, providing actionable advice and real-world examples to address common user pain points.
If you've ever wondered about what the molecular mass of a compound is, you're not alone. Many people find this concept daunting due to its seemingly complex calculations. However, once broken down step-by-step, it's straightforward and immensely useful in your chemistry studies or professional work. This guide will walk you through the essentials of determining the molecular mass of compounds, with a particular focus on carbon. Let's get started by addressing the core problem and offering a practical solution for understanding this fundamental concept.
The Problem of Understanding Molecular Mass
Determining the molecular mass, also known as the molecular weight, of a compound can be a complex task, especially for beginners in chemistry. The confusion typically arises from the need to sum up the atomic masses of all atoms in a molecule, which seems daunting without a clear method. For instance, you might be asked to find the molecular mass of a common compound like glucose (C6H12O6). The sheer number of atoms and their atomic masses can make this task intimidating. Moreover, many students struggle to apply this concept to real-world chemical equations and lab work. Understanding molecular mass is key to mastering stoichiometry and making accurate predictions about chemical reactions.
Here’s where we simplify the process, providing you with a step-by-step guide to determine the molecular mass of any compound, with a particular emphasis on those containing carbon. By breaking down the steps and offering practical tips and examples, we aim to alleviate the confusion and make this concept accessible and useful.
Quick Reference
Quick Reference
- Immediate action item with clear benefit: Use a periodic table to find the atomic mass of each element in your compound.
- Essential tip with step-by-step guidance: Write down the molecular formula, identify each atom, multiply the number of each type of atom by its atomic mass, and then sum the results.
- Common mistake to avoid with solution: Forgetting to count each atom’s number in the formula. Solution: Always double-check the subscripts in the chemical formula.
How to Determine the Molecular Mass of a Compound Containing Carbon
Let’s dive into the details of calculating the molecular mass of a compound with carbon. The process is systematic and, once you get the hang of it, quite manageable. Below, we’ll walk through an example using glucose (C6H12O6), a compound you’re likely familiar with due to its role in biological systems.
Step 1: Identify the Compound
The first step is to clearly identify the compound you are working with. In this case, we’ll use glucose (C6H12O6). The molecular formula provides the counts of each type of atom in the molecule.
Step 2: Gather Atomic Masses
Next, gather the atomic masses of each element involved. You can find these on a periodic table. The atomic masses are usually listed below the element’s symbol. For example, carbon © has an atomic mass of approximately 12.01 amu (atomic mass units), hydrogen (H) has 1.008 amu, and oxygen (O) has 16.00 amu.
Step 3: Calculate the Total Mass for Each Element
Multiply the atomic mass of each element by the number of atoms of that element in the molecule. For glucose:
- Carbon ©: 6 atoms × 12.01 amu = 72.06 amu
- Hydrogen (H): 12 atoms × 1.008 amu = 12.096 amu
- Oxygen (O): 6 atoms × 16.00 amu = 96.00 amu
Step 4: Sum the Results
Finally, sum the total masses from each element to find the molecular mass of the compound. For glucose:
72.06 amu © + 12.096 amu (H) + 96.00 amu (O) = 180.156 amu
So, the molecular mass of glucose is approximately 180.156 amu. This method can be applied to any compound, as long as you correctly identify the elements and their quantities in the molecular formula.
Practical Example: Ethylene Glycol (C2H6O2)
Let’s walk through another example, ethylene glycol (C2H6O2), which is a common antifreeze component. Follow the same steps:
- Identify the compound: Ethylene glycol (C2H6O2).
- Gather atomic masses: Carbon © = 12.01 amu, Hydrogen (H) = 1.008 amu, Oxygen (O) = 16.00 amu.
- Calculate the total mass for each element: 2 × 12.01 amu © = 24.02 amu, 6 × 1.008 amu (H) = 6.048 amu, 2 × 16.00 amu (O) = 32.00 amu.
- Sum the results: 24.02 amu © + 6.048 amu (H) + 32.00 amu (O) = 62.068 amu.
Therefore, the molecular mass of ethylene glycol is approximately 62.068 amu.
FAQ Section
What if the compound has a complex molecular formula?
For complex molecular formulas, the same steps apply. Identify each type of atom and count them carefully. For example, in vitamin C (C6H8O6), you would calculate as follows:
- Carbon ©: 6 atoms × 12.01 amu = 72.06 amu
- Hydrogen (H): 8 atoms × 1.008 amu = 8.064 amu
- Oxygen (O): 6 atoms × 16.00 amu = 96.00 amu
Summing these: 72.06 amu © + 8.064 amu (H) + 96.00 amu (O) = 176.124 amu. Thus, the molecular mass of vitamin C is approximately 176.124 amu.
Tips and Best Practices
Here are some tips to ensure you correctly determine the molecular mass:
- Accuracy: Always use the most accurate atomic masses available from the periodic table.
- Double-check: Re-check the molecular formula to ensure you have not missed any atoms.
- Practice: Work through several examples to become more comfortable with the process.
- Use a calculator: For large numbers, it’s easy