Precenting at the molecular level, methanol, a small yet mighty compound, serves as a fundamental cornerstone in both industrial and biological arenas. With its molecular formula, CH₃OH, methanol exhibits properties that render it pivotal in numerous applications, from antifreeze in vehicles to its role as a precursor in chemical synthesis. This article delves into the molecular weight of methanol, underscoring its significance and applications.
Understanding the Molecular Weight of Methanol
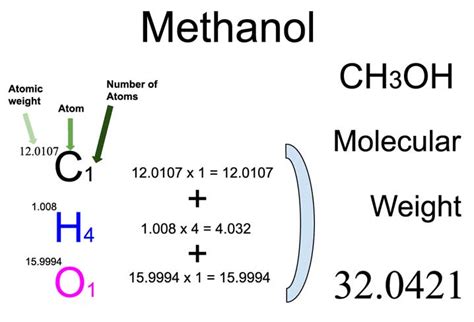
Methanol, known chemically as methyl alcohol or wood alcohol, has a molecular weight of approximately 32.04 g/mol. This measurement is pivotal as it helps in understanding the behavior of methanol in chemical reactions and its interactions with other substances. For example, in industrial processes, knowing the precise molecular weight allows for accurate dosage and efficient utilization in chemical reactions, minimizing waste and enhancing productivity.
Key Insights
- The molecular weight of methanol is essential for its application in chemical reactions.
- Understanding methanol’s molecular weight aids in the optimization of industrial processes.
- Proper handling based on molecular weight helps prevent environmental and health hazards.
Applications in Industry
In the industrial sector, methanol is utilized in diverse processes such as the production of formaldehyde, which is a key ingredient in many household products including resins, plastics, and adhesives. Moreover, methanol serves as a building block in creating dimethyl ether (DME), a clean-burning fuel that can be used in engines and as a solvent in various industries.
Methanol’s versatility stems from its relatively low molecular weight, which allows it to interact readily with other compounds, facilitating its use in both chemical synthesis and as a fuel additive. Its presence in gas-to-liquid (GTL) technologies highlights its role in advancing sustainable energy solutions by producing cleaner fuels.
Role in Biology and Environment
On a biological level, methanol plays a crucial role in the metabolic processes of certain microorganisms and plants. In humans, ingestion of methanol, however, is toxic and requires prompt medical intervention. The liver metabolizes methanol into formaldehyde and formic acid, both of which are highly toxic substances that can lead to severe health issues, including blindness and death if not promptly addressed.
Understanding methanol’s molecular weight and its biological impact is critical for environmental safety and health. The detoxification process mediated by enzymes such as alcohol dehydrogenase (ADH) is a testament to the body’s intricate defense mechanisms against such toxic substances. Knowledge of methanol’s weight aids in designing better antidotes and treatment protocols.
Is methanol environmentally safe?
Methanol is not inherently environmentally safe. It is toxic to aquatic life and can pose significant health risks to humans. However, its role in sustainable energy initiatives like GTL technologies shows promise in reducing overall environmental impact when used and managed correctly.
What happens if methanol is ingested?
Ingesting methanol leads to rapid metabolism into toxic substances such as formaldehyde and formic acid, which can cause severe health issues including permanent blindness and death. Immediate medical intervention is crucial.
This article has outlined the importance of understanding methanol’s molecular weight, emphasizing its diverse applications in industry and its critical role in biological and environmental contexts. As we advance in both technology and scientific knowledge, leveraging such molecular insights will be imperative in optimizing processes, ensuring safety, and advancing sustainable practices.