Mastering NF3 Lewis Structure: Unlock Chemistry Secrets Today!
Understanding the NF3 Lewis structure is a fundamental step towards grasping more complex topics in chemistry. This guide will walk you through the step-by-step process of drawing the Lewis structure for NF3, ensuring you understand not only the process but also the reasoning behind each move. Whether you are a student preparing for an exam, a chemist seeking to deepen your knowledge, or just a curious mind, this guide provides practical examples and expert tips to demystify this concept.
Why NF3 Lewis Structure Matters
The NF3 molecule is a cornerstone in understanding how valence electrons are shared and bonded in covalent molecules. By mastering the NF3 Lewis structure, you unlock insights into molecular geometry, polarity, and reactivity, all of which are crucial for advanced studies in chemistry. A solid grasp of this will empower you to predict how other molecules might behave and interact.
Immediate Action: A Quick Start
Before diving into the intricate details, let’s address the most immediate and impactful steps:
Quick Reference
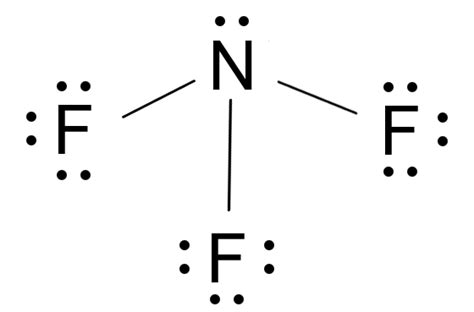
- Begin by counting valence electrons: Nitrogen has 5 and Fluorine has 7; NF3 has 26 electrons in total.
- Place Nitrogen at the center and connect it to three Fluorines with single bonds.
- Distribute the remaining electrons to satisfy the octet rule, avoiding common pitfalls like incorrect bond placements.
Drawing the NF3 Lewis Structure: Step-by-Step
Drawing the Lewis structure for NF3 involves several clear, methodical steps. This section will guide you through each phase, ensuring you fully comprehend the process.
Step 1: Count Total Valence Electrons
Start by determining the total number of valence electrons in the NF3 molecule. Nitrogen (N) has 5 valence electrons, and each Fluorine (F) has 7. Since there are three Fluorines, you multiply 7 by 3 to get 21. Add these together with Nitrogen's 5 valence electrons, resulting in a total of 26 valence electrons.
Step 2: Place Nitrogen at the Center
Nitrogen is less electronegative compared to Fluorine, so it should be the central atom. Place Nitrogen at the center of your sketch and draw the three Fluorines around it, ensuring a symmetrical structure.
Step 3: Connect with Single Bonds
Next, draw single bonds from the Nitrogen atom to each of the three Fluorine atoms. Each bond uses up 2 electrons, so three bonds account for 6 electrons. This leaves 20 valence electrons to distribute.
Step 4: Distribute Remaining Electrons
Now, place the remaining electrons on the Fluorines to satisfy their octet rule. Each Fluorine requires 1 more pair of electrons to complete an octet. Distribute these electrons one pair at a time to each Fluorine atom.
Step 5: Complete Nitrogen’s Octet
At this stage, Nitrogen should have only 8 electrons in its valence shell, which is satisfied by the 6 electrons from the bonds and 2 additional electrons placed directly on it.
Detailed How-To Sections
Advanced Tips for Drawing Lewis Structures
Here are some expert tips to refine your approach and avoid common mistakes:
- Ensure that you always place the least electronegative atom at the center of your structure.
- Double-check each atom’s octet rule compliance; often, checking your work at intermediate steps can prevent errors.
- Avoid over-complicating your drawing; keep it simple and focus on the rules first.
Common Mistakes to Avoid
Here are some common mistakes and their solutions:
- Incorrect Bond Placement: Always place bonds correctly. In NF3, Nitrogen should be central with Fluorines surrounding it.
- Over-filling Octets: Never allow an atom to have more than 8 electrons unless it’s in the third period or beyond and is specifically stable with 10 or 12 electrons (rare).
- Electron Neglect: Don’t forget to distribute all electrons properly; it’s easy to miss a Fluorine needing additional pairs.
Practical FAQ
What is the molecular geometry of NF3?
To determine the molecular geometry of NF3, we need to consider the arrangement of atoms and lone pairs around the central atom. Nitrogen has three single bonds to Fluorines and one lone pair of electrons. According to VSEPR (Valence Shell Electron Pair Repulsion) theory, this configuration results in a trigonal pyramidal molecular geometry. The presence of the lone pair slightly distorts the perfect tetrahedron shape, making it pyramidal.
Is NF3 polar or non-polar?
To determine the polarity of NF3, consider both the molecular geometry and the electronegativity difference between Nitrogen and Fluorine. Although the NF3 molecule is trigonal pyramidal in shape, the lone pair on Nitrogen causes an asymmetrical distribution of electron density, resulting in a net dipole moment. Since Fluorine is more electronegative than Nitrogen, the pull on the shared electron pairs is uneven, making NF3 a polar molecule. This polarity influences its physical properties, such as its boiling and melting points, and its interactions with other molecules.
Conclusion
Mastering the NF3 Lewis structure involves a blend of understanding electron distribution, molecular geometry, and polarity. By following the steps outlined in this guide, you should be well-equipped to draw and analyze the NF3 structure. Remember to check your work regularly to ensure accuracy and avoid common pitfalls. With practice, these steps will become second nature, allowing you to tackle more complex molecules with confidence.