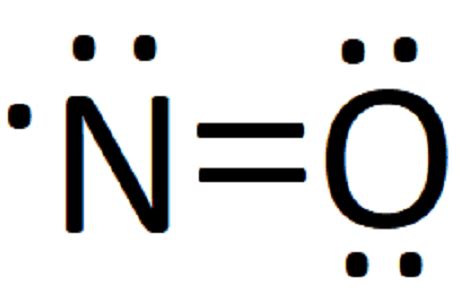If you've ever felt overwhelmed by the complexity of chemistry and especially Lewis structures, you’re not alone. Many students find traditional methods challenging and it can be easy to feel lost. Luckily, there’s an alternative approach that focuses more on understanding chemical reactions and molecular behavior without delving into the intricate details of Lewis structures. This guide will help you navigate this simplified path with actionable steps, real-world examples, and practical advice. We'll demystify the process, provide quick tips, and answer common questions, ensuring you can apply these methods effectively.
Understanding the Problem: Why Traditional Methods Can Be Overwhelming
Traditional chemistry education often emphasizes detailed Lewis structures, which involve drawing complex diagrams to represent electrons around atoms. While this method has its merits, it can be an unnecessary hurdle for many learners, especially those new to the subject. The focus shifts from understanding the chemistry to mastering the art of diagram-drawing, often leaving little room for deeper conceptual understanding. The challenge here is that many students, while capable, become intimidated or confused by the level of detail required, leading to frustration and a potential dislike for chemistry.
How Traditional Methods Fail
The traditional approach often fails by:
- Requiring excessive memorization of rules and guidelines for Lewis structures.
- Over-complicating the learning process with unnecessary detail.
- Leaving little room for conceptual exploration and understanding.
The Solution: A Simplified Approach to Chemistry
By focusing on reaction mechanisms, molecular behavior, and physical properties rather than memorizing Lewis structures, we can make chemistry more accessible and engaging. This approach emphasizes the 'why' behind chemical reactions, offering a more intuitive understanding. This guide will walk you through the basics of this simplified method, providing you with the tools to grasp key concepts without the need for complex diagrams.
Immediate Action: Start with Simple Concepts
Begin by focusing on the most basic principles of chemistry: atoms, molecules, and the basics of chemical bonding. Here’s what you should do right away:
- Identify and understand the most common types of chemical bonds.
- Learn the basics of how atoms combine to form molecules.
- Understand the concept of valence electrons and how they influence molecular structure.
Essential Tip: Visualize Simple Molecules
Instead of trying to draw detailed Lewis structures, start by visualizing simple molecules like water (H2O) or carbon dioxide (CO2). Use basic shapes and consider how atoms bond together based on their electron needs.
Common Mistake to Avoid: Over-complicating Simple Ideas
Don’t try to apply complex rules or structures that you haven’t yet mastered. Keep your initial focus on basic concepts and avoid diving into the intricacies of Lewis structures until you're more comfortable.
How to Master the Basics
Understanding the basics of chemistry without using Lewis structures involves understanding how atoms form bonds and how molecules interact. Here's a step-by-step guide to get you started.
Step 1: Understanding Atomic Structure
Every atom is made up of protons, neutrons, and electrons. The number of protons determines the element’s identity and is shown by its atomic number. For most chemical reactions, it’s crucial to focus on the outermost electrons, known as valence electrons. These electrons are involved in forming bonds and are the primary concern in our simplified approach.
Step 2: Learning Basic Types of Bonds
Atoms combine to form molecules through various types of bonds. Here are the basics:
- Covalent Bonds: These involve atoms sharing pairs of electrons. Think of it as a 'handshake' between atoms. For example, in a water molecule (H2O), oxygen shares electrons with each hydrogen atom.
- Ionic Bonds: These occur when one atom donates an electron to another. This typically happens between metals and non-metals. For example, in sodium chloride (NaCl), sodium donates an electron to chlorine, forming ions that attract each other.
Step 3: Visualizing Molecules
Instead of drawing complex structures, begin by visualizing how atoms might bond. Here’s a practical approach:
- Start with simple molecules: Begin with molecules like water (H2O), methane (CH4), or carbon dioxide (CO2). Consider what kind of bond they might form.
- Use simple diagrams: Draw circles to represent atoms and lines to indicate bonds. For water, represent oxygen with two lines (each line representing a bond to two hydrogen atoms).
- Think about electron needs: Remember, atoms form bonds to achieve stability, often trying to have a full outer shell of electrons. Oxygen, for example, needs two more electrons to fill its outer shell, so it bonds with two hydrogens each needing one electron to fill their shells.
FAQ: Common Questions About the No Lewis Structure Approach
Will I still understand complex molecules without Lewis structures?
Absolutely. The key here is focusing on the principles of bonding and molecular behavior. Rather than memorizing complex structures, you will learn how atoms combine based on their electronic configurations and the rules governing those combinations. This foundational understanding allows you to grasp the basics of more complex molecules.
What if I still need to draw Lewis structures for exams?
If exams require Lewis structures, it’s best to practice selectively. Start by learning the essential rules that govern bond formation and electron sharing. Focus on understanding when and why certain structures are formed rather than rote memorization. If you need to draw one, use your simplified understanding as a guide—think in terms of basic bonding principles rather than complex diagrams.
Can this approach help me with reaction mechanisms?
Yes, focusing on molecular behavior rather than structures can actually enhance your understanding of reaction mechanisms. Instead of focusing on how atoms are arranged in a molecule, concentrate on why certain reactions occur. For example, understand how molecules interact to form new compounds based on their electron needs and bonding types. This approach makes learning about reaction mechanisms more intuitive and less daunting.
By adopting this simplified approach, you can gain a solid grasp of fundamental chemistry concepts without getting bogged down by the complexities of Lewis structures. This method makes the subject more approachable and enjoyable, paving the way for deeper understanding and more advanced study in chemistry.