Understanding the molar mass of octane is critical for anyone diving into chemistry, whether you’re a student, a professional, or just an enthusiast. Many find the concept daunting due to the complexities involved in understanding chemical formulas, molecular weights, and periodic table applications. This guide is your gateway to simplifying and mastering the molar mass calculation for octane, a fundamental topic in chemistry.
To begin, the molar mass of a substance, including octane, refers to the mass of one mole of that substance—typically measured in grams per mole (g/mol). For octane, a commonly used hydrocarbon with the chemical formula C8H18, calculating its molar mass involves understanding each atom's contribution. This guide walks you through the process, provides quick reference tips, and offers a comprehensive FAQ to address common questions.
Unlocking the Mystery: Why Molar Mass Matters
Molar mass is essential in chemistry because it provides a bridge between the atomic scale and the macroscopic scale, allowing chemists to measure quantities in reactions, predict outcomes, and understand the composition of various substances. For octane, which is significant in fuels and petrochemical industries, knowing its molar mass is crucial for various practical applications, from lab calculations to industrial processes. Grasping this concept allows for accurate formulation, reaction prediction, and material composition analysis.
Quick Reference Guide
Quick Reference
- Immediate action item: Identify the molecular formula of octane (C8H18).
- Essential tip: Use the periodic table to find atomic masses for carbon © and hydrogen (H).
- Common mistake to avoid: Confusing the number of atoms with their atomic masses, leading to incorrect molar mass calculations.
Detailed Guide to Calculating Octane’s Molar Mass
Let’s dive deeper into the process of calculating the molar mass of octane. This will be broken down into simple, easy-to-follow steps that anyone can understand, whether they’re a beginner or just need a refresher.
Step-by-Step Calculation
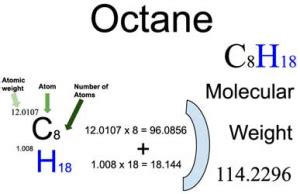
The molar mass of octane, C8H18, is calculated by adding together the molar masses of its constituent atoms. Here’s how you can do it:
- Determine the atomic masses: Look up the atomic mass of each element on the periodic table. The atomic mass for carbon (C) is approximately 12.01 g/mol, and for hydrogen (H), it is approximately 1.01 g/mol.
- Count the atoms: In the formula C8H18, there are 8 carbon atoms and 18 hydrogen atoms.
- Calculate the total molar mass: Multiply the atomic mass of each element by the number of its respective atoms in the formula and then add them together.
Here’s the formula breakdown:
| Element | Atomic Mass (g/mol) | Number of Atoms | Total Contribution (g/mol) |
|---|---|---|---|
| Carbon (C) | 12.01 | 8 | 8 * 12.01 = 96.08 g/mol |
| Hydrogen (H) | 1.01 | 18 | 18 * 1.01 = 18.18 g/mol |
Adding these together gives you:
96.08 g/mol (carbon) + 18.18 g/mol (hydrogen) = 114.26 g/mol
Practical Application and Examples
Let’s put this theory into practice with some real-world examples:
Imagine you’re working in a lab and need to prepare a solution that requires 0.5 moles of octane. To find out how many grams you need, you would first calculate the molar mass of octane (114.26 g/mol) and then use the following formula:
mass = moles × molar mass
So for 0.5 moles of octane:
0.5 moles * 114.26 g/mol = 57.13 grams
Advanced Tips and Best Practices
To further refine your understanding and application of octane’s molar mass, consider these advanced tips:
- Use a calculator: For larger numbers and more complex calculations, a scientific calculator can help you manage precision and speed.
- Practice with different compounds: Once comfortable with octane, try calculating molar masses for other compounds to build confidence and understanding.
- Review the periodic table: Regularly update your knowledge of atomic masses from the periodic table, as values might slightly vary across different references.
Practical FAQ Section
What is the difference between molar mass and molecular mass?
Molar mass and molecular mass are essentially the same concept but used in different contexts. Molecular mass refers to the mass of a single molecule of a compound, often expressed in atomic mass units (amu). Molar mass, however, is this same concept expressed in grams per mole (g/mol), and it applies to one mole of any substance, whether it’s a molecule, an atom, or a formula unit. For example, the molecular mass of octane (C8H18) can be found by summing the atomic masses of its constituent atoms, but its molar mass, which chemists more often use, is the same calculation expressed in g/mol.
How do you calculate the molar mass of complex compounds?
To calculate the molar mass of more complex compounds, follow the same principle: sum the molar masses of all atoms in the formula. For instance, if you’re working with ethanol (C2H6O), you first find the atomic masses for each element (carbon: 12.01 g/mol, hydrogen: 1.01 g/mol, oxygen: 16.00 g/mol) and then multiply these by the number of each atom present in the formula.
So for ethanol:
- Carbon: 2 atoms * 12.01 g/mol = 24.02 g/mol
- Hydrogen: 6 atoms * 1.01 g/mol = 6.06 g/mol
- Oxygen: 1 atom * 16.00 g/mol = 16.00 g/mol
Adding these gives a total molar mass of 46.08 g/mol.
Understanding the molar mass of octane or any compound is foundational in chemistry. By mastering these calculations, you’ll find that the world of chemical reactions, formulations, and analyses opens up to a greater degree of precision and insight. Keep practicing, stay curious, and let the intricacies of chemistry unveil themselves to you!