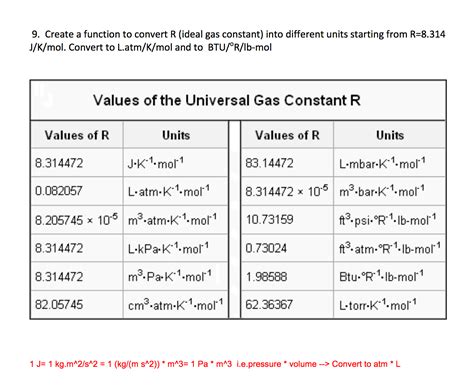
The gas constant, often represented as ‘R’, plays an integral role in advanced chemistry, serving as a cornerstone in various gas laws and equations. Its mastery can unlock deeper understandings of thermodynamic processes, allowing for precise calculations in fields ranging from industrial chemistry to environmental science. This article delves into the essentials of the gas constant, providing expert insights, evidence-based examples, and practical applications to enhance your proficiency.
Understanding the Universal Gas Constant
In the realm of gases, the universal gas constant ® is a crucial parameter, appearing prominently in the ideal gas law equation: PV=nRT. This constant quantifies the energy related to a specific amount of a gas at a specified temperature. R’s significance lies in its uniformity across all gases, making it a pivotal tool in comparing different gases under various conditions.
Key Insights
- Primary insight with practical relevance: Understanding 'R' helps in predicting the behavior of gases under varying temperature and pressure.
- Technical consideration with clear application: Knowing 'R' aids in accurate energy calculations in thermodynamic cycles.
- Actionable recommendation: Incorporate 'R' in real-world applications, such as engineering projects or environmental modeling.
Application of the Gas Constant in Thermodynamic Equations
The utility of the gas constant becomes particularly evident in the analysis of thermodynamic processes. For example, consider the application of ‘R’ in the calculation of work done by or on a gas during isothermal expansion. The work done, W, in an isothermal process is given by W=nRTln(V2/V1). Here, ‘R’ directly influences the outcome, making its precise knowledge imperative for accurate assessments.
Moreover, ‘R’ is essential in determining gas molar volume. Using the ideal gas law (PV=nRT), engineers can determine the volume occupied by a specific number of moles of gas at given pressure and temperature conditions, which is vital in designing and optimizing industrial processes.
Environmental and Industrial Implications
In the environmental sector, ‘R’ plays a significant role in modeling atmospheric processes and climate systems. For instance, the ideal gas law is applied to estimate the amount of greenhouse gases in the atmosphere. Accurate predictions of gas concentrations are crucial for understanding climate change dynamics and developing mitigation strategies.
Industrially, ‘R’ is employed in fields like chemical engineering, where it helps in the design and operation of reactors and separation units. By understanding gas behavior under different conditions, engineers can optimize processes for higher efficiency and lower environmental impact.
What is the value of the universal gas constant 'R'?
The value of 'R' is approximately 8.314 J/(mol·K). This constant is used in various gas law equations to relate the pressure, volume, and temperature of a gas.
How is 'R' used in real-world applications?
'R' is used in applications ranging from calculating the work done by gases in thermodynamic cycles to predicting atmospheric conditions in climate models. It is a fundamental constant in understanding and predicting gas behavior.
In conclusion, mastering the gas constant ‘R’ opens doors to advanced understanding and application across diverse fields of chemistry and engineering. Its accurate use enables precise predictions and optimizations, making it an indispensable tool in both theoretical and practical contexts. With this comprehensive insight, you are now equipped to unlock the secrets of ‘R’ and apply this knowledge to your professional endeavors.