In the realm of chemistry, single replacement displacement reactions are fundamental yet intricate processes that warrant detailed exploration. These reactions play a pivotal role in various scientific and industrial applications, and understanding their nuances can significantly enhance your grasp of chemical principles.
Key insights box:
Key Insights
- Primary insight with practical relevance: Mastery of single replacement displacement reactions aids in developing problem-solving skills for more complex chemical processes.
- Technical consideration with clear application: Predicting the outcomes of these reactions requires an understanding of reactivity series and the properties of the involved elements.
- Actionable recommendation: Regularly practice identifying and predicting single replacement reactions to reinforce your comprehension and application of chemical principles.
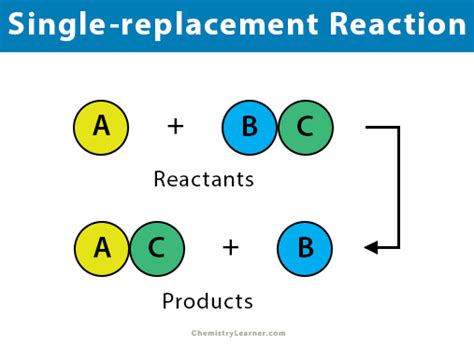
Single replacement displacement reactions, also known as single displacement reactions, occur when one element replaces another in a compound. This phenomenon can be depicted through the general formula:
A + BC -> B + AC
Here, element A displaces element B from compound BC, forming compound AC. This type of reaction occurs under specific conditions, primarily governed by the reactivity of the elements involved.
In these reactions, the reactivity of the elements determines the feasibility and spontaneity. More reactive elements have a greater tendency to displace less reactive elements. For instance, when zinc (Zn) reacts with hydrochloric acid (HCl), zinc displaces hydrogen (H) because zinc is more reactive than hydrogen.
The equation for this reaction would look like:
Zn + 2HCl -> H2 + ZnCl2
This reaction exemplifies the practical relevance of understanding single replacement displacement reactions. Here, zinc displaces hydrogen from hydrochloric acid, producing hydrogen gas and zinc chloride.
Another example is the reaction between magnesium (Mg) and copper sulfate (CuSO4). Magnesium, being more reactive than copper, displaces copper from its sulfate compound to form magnesium sulfate (MgSO4) and copper (Cu).
The balanced equation is:
Mg + CuSO4 -> Cu + MgSO4
In this case, the displacement reaction underscores the importance of the reactivity series in predicting the outcome of the reaction.
Two analysis sections:
Reactivity Series and Its Application
A crucial component in analyzing single replacement displacement reactions is the reactivity series. This series ranks metals based on their ability to lose electrons and form positive ions. The higher a metal is on the series, the more reactive it is. For instance, metals higher than hydrogen in the series can displace hydrogen from acids, thereby forming hydrogen gas and a salt.
Understanding the reactivity series allows chemists to predict the likelihood of a single replacement displacement reaction occurring. For instance, if we consider the metals potassium (K), sodium (Na), calcium (Ca), magnesium (Mg), and aluminum (Al), we can ascertain that potassium will displace sodium from sodium chloride (NaCl), whereas sodium cannot displace potassium from potassium chloride (KCl) because potassium is higher in the reactivity series.
Environmental and Industrial Implications
Single replacement displacement reactions have significant environmental and industrial implications. In environmental chemistry, these reactions are vital in understanding natural processes such as weathering of minerals and corrosion of metals. For example, the corrosion of iron (Fe) in the presence of water and oxygen is a single replacement displacement reaction where iron displaces hydrogen in water to form iron oxide (rust).
Industrially, these reactions are harnessed in various processes, including the extraction of metals from their ores. For instance, in the extraction of zinc from zinc sulfide (ZnS), carbon (C) displaces sulfur (S), forming zinc metal and sulfur dioxide (SO2). This reaction is crucial in metallurgy and highlights the industrial application of single replacement displacement reactions.
FAQ section:
What makes an element a good candidate for displacement in a single replacement reaction?
An element that is higher in the reactivity series is a better candidate for displacement in a single replacement reaction because it has a greater tendency to lose electrons and replace another element in a compound.
Are there any safety precautions to consider when conducting these reactions?
Yes, certain safety precautions should be followed, particularly when dealing with reactive metals or acids. Always use protective equipment, ensure proper ventilation, and follow standard laboratory safety protocols to mitigate risks associated with these reactions.
Understanding single replacement displacement reactions not only solidifies your foundational knowledge of chemistry but also opens doors to advanced applications in both scientific research and industrial processes. With these insights and examples, you are better equipped to appreciate and apply the principles of single replacement displacement reactions in practical scenarios.