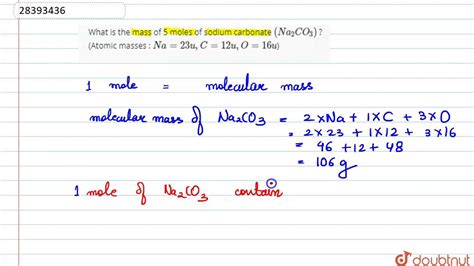The determination of sodium carbonate molar mass is an essential component in the field of chemistry. This fundamental concept is frequently utilized in various laboratory and industrial settings. Knowing the molar mass is critical for accurate stoichiometric calculations, which are the backbone of chemical analysis and synthesis. This article aims to provide a concise yet comprehensive guide to sodium carbonate molar mass, focusing on expert perspectives, practical insights, and real-world examples.
Key Insights
- Primary insight with practical relevance: Understanding the molar mass of sodium carbonate, a key compound in cleaning agents and chemical manufacturing, is vital for precise chemical reactions and formulations.
- Technical consideration with clear application: Sodium carbonate’s molar mass, determined through elemental analysis, allows chemists to perform accurate molarity calculations essential for experimental procedures.
- Actionable recommendation: Always verify the molar mass using updated chemical data resources to ensure accuracy in your calculations and chemical processes.
Understanding Sodium Carbonate Molar Mass
Sodium carbonate (Na2CO3), commonly known as soda ash or washing soda, is a versatile compound with extensive applications in industry and laboratory settings. The molar mass is the sum of the atomic masses of all atoms in the formula. This involves sodium (Na), carbon ©, and oxygen (O). Sodium carbonate contains two sodium atoms, one carbon atom, and three oxygen atoms. Calculating the molar mass requires knowledge of the atomic weights of these elements: approximately 23 g/mol for sodium, 12 g/mol for carbon, and 16 g/mol for oxygen. The calculation is as follows: 2(23) + 12 + 3(16) = 106 g/mol. Therefore, the molar mass of sodium carbonate is precisely 106 g/mol.Applications of Sodium Carbonate in Various Industries
The significance of sodium carbonate molar mass extends beyond basic chemistry into numerous industrial applications. In the manufacturing sector, soda ash is crucial in producing glass, paper, detergents, and other chemicals. Accurate determination of molar mass is essential for precise formulation and quality control in these products. For instance, the detergent industry relies on exact stoichiometric measurements to balance the chemical composition of washing powders, ensuring effective cleaning while minimizing environmental impact.Experimental Implications
In laboratory settings, sodium carbonate’s molar mass is indispensable for accurate experimental calculations. Whether titrating a solution to determine acidity or conducting reactions involving carbonate salts, precise molar mass values are necessary to maintain the integrity of experimental results. For example, when preparing a sodium carbonate solution for a neutralization reaction, knowing its molar mass ensures that the concentration of the solution is correctly established, thus facilitating accurate prediction and measurement of the reaction’s outcome.Is the molar mass of sodium carbonate always the same?
Yes, the molar mass of sodium carbonate is a fixed value, 106 g/mol, based on the atomic weights of sodium, carbon, and oxygen. However, any impurity or isotopic variation in the sample can affect its actual mass.
How does knowing the molar mass of sodium carbonate help in industrial applications?
Understanding the molar mass allows for precise quantification and formulation, critical for quality control and product consistency in industries such as glass manufacturing, detergent production, and chemical synthesis.
In summary, the knowledge of sodium carbonate molar mass is integral to both academic and industrial chemistry. From precise laboratory experiments to large-scale industrial applications, accurate molar mass determination ensures the successful execution of chemical processes and product formulations.