Are you curious about how synthesis reactions work in real life and want practical examples that you can implement? This guide is crafted to provide you with step-by-step guidance, actionable advice, and real-world solutions to understand synthesis reactions better. Whether you are a student, a professional in a related field, or simply someone fascinated by chemical reactions, this guide will address your pain points and equip you with the knowledge to master synthesis reactions.
Understanding Synthesis Reactions: The Basics
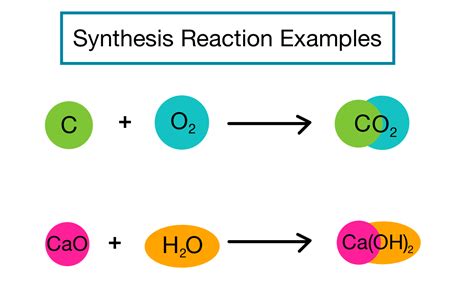
A synthesis reaction, often called a combination reaction, occurs when two or more simple substances combine to form a more complex product. It’s a fundamental concept in chemistry that finds applications in everyday life, from manufacturing various products to environmental chemistry.
To help you get a quick grasp, here’s a quick reference guide:
Quick Reference
- Immediate action item: Watch a live demonstration of a synthesis reaction to better visualize the process.
- Essential tip: Always ensure you have proper safety gear and understand the chemical properties of the reactants.
- Common mistake to avoid: Not checking for any impurities in your reactants which can lead to unexpected results.
Step-by-Step Guide to Conducting a Synthesis Reaction
Synthesis reactions can vary greatly, but here we’ll delve into a practical example that is commonly used in educational and industrial settings: the synthesis of magnesium oxide from magnesium metal and oxygen.
The chemical equation for this reaction is:
2Mg(s) + O2(g) → 2MgO(s)
This reaction highlights the basic principles and allows us to explore the steps involved comprehensively.
Step 1: Preparation of Reactants
Begin by ensuring you have high-purity magnesium strips and oxygen gas. Impurities can interfere with the reaction, so use laboratory-grade materials.
Here’s a list of materials you will need:
- Magnesium strips
- Oxygen gas supply
- Ignition tool (e.g., a magnesium igniter or a Bunsen burner)
- Safety equipment (gloves, goggles, lab coat)
- Heat-resistant container (e.g., a ceramic or metal crucible)
Step 2: Setting Up the Reaction
Place the magnesium strip inside the heat-resistant container. Ensure the container is stable and placed on a heat source, such as a Bunsen burner.
Step 3: Initiating the Reaction
Under safe conditions, use the ignition tool to light the magnesium strip. As the magnesium burns, it will produce bright light, and the reaction with oxygen will form magnesium oxide. Monitor the reaction to ensure it proceeds smoothly.
This step is crucial because magnesium reacts vigorously with oxygen. Proper ventilation and safety gear are essential to prevent accidents.
Step 4: Observing the Results
Once the reaction completes, carefully extinguish the magnesium strip’s flame and allow the magnesium oxide to cool. Observe the transformation from magnesium metal to magnesium oxide.
This transformation is not just a visual change but also a significant chemical change as the magnesium and oxygen atoms rearrange to form a new compound.
Step 5: Cleanup and Analysis
After ensuring everything has cooled, carefully clean up the workspace. Document the results and any observations for further analysis.
Weigh the magnesium oxide formed to calculate the yield and compare it with theoretical values to understand the efficiency of the reaction.
Advanced Tips for Mastery
For those looking to advance their understanding, here are some tips:
Consider conducting the reaction under varying conditions, such as different temperatures or pressures, to observe how these factors influence the reaction rate and product yield.
Also, explore other synthesis reactions like the creation of water from hydrogen and oxygen, a reaction that is fundamental to both natural processes and industrial applications.
Practical FAQ
What safety precautions should I take when performing synthesis reactions?
Safety should always be your top priority. Here’s a detailed step-by-step guide to ensure your safety:
- Wear appropriate personal protective equipment (PPE) including gloves, goggles, and lab coats.
- Conduct reactions in a well-ventilated area or under a fume hood to avoid inhalation of any harmful gases.
- Always have fire extinguishing equipment nearby in case of accidents.
- Be familiar with the specific safety data sheets (SDS) for all chemicals used.
- Never perform reactions without proper supervision, especially if you are a beginner.
These guidelines are designed to give you confidence and understanding as you explore synthesis reactions. With practical examples and real-world applications, you can appreciate the depth and utility of these chemical processes.
Remember, the key to mastering synthesis reactions lies in repeated practice, careful observation, and thorough understanding of both the theory and practical application. This guide aims to provide that foundational knowledge and the tools you need to succeed.