Understanding the Freezing Point Fahrenheit is essential for various scientific and practical applications. This concept, often misunderstood, plays a crucial role in meteorology, culinary arts, and material sciences. This article delves into the significance of the freezing point in Fahrenheit and provides expert insights grounded in evidence and real-world examples.
Significance of Freezing Point Fahrenheit
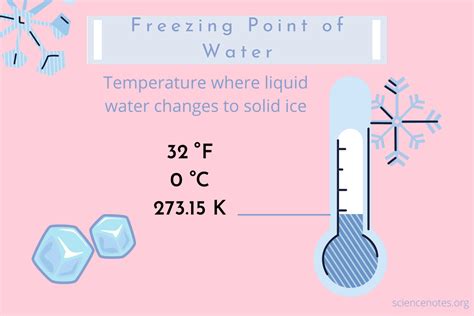
The freezing point of a substance, the temperature at which it transitions from liquid to solid, is a fundamental property with broad implications. In Fahrenheit, the freezing point of water is 32°F, a benchmark often utilized in weather forecasting, cooking, and even in determining the quality of materials used in construction. This ubiquitous temperature is pivotal for predicting frost and freezing conditions, aiding in timely preparation and prevention of damage.
Key Insights
Key Insights
- The freezing point of water at 32°F serves as a critical indicator in meteorology.
- In culinary arts, knowing the freezing point can help in achieving the perfect texture for ice cream and frozen desserts.
- Understanding this concept aids in selecting appropriate materials for construction in cold climates.
Meteorological Implications
In meteorology, the freezing point is a pivotal factor for forecasting weather patterns and warning systems. Temperatures at or below 32°F can lead to freezing rain, sleet, or even snow, all of which can have significant impacts on transportation, infrastructure, and daily life. For example, in regions where the average winter temperature hovers just above or below this critical mark, weather agencies meticulously monitor these conditions to predict and manage potential disruptions such as icy roads, flight cancellations, and power outages.
Culinary and Material Science Applications
Beyond its meteorological applications, the freezing point of 32°F is instrumental in the culinary arts and material sciences. In cooking, especially for dessert preparation, the precise knowledge of this temperature ensures that ingredients like ice cream, sorbets, and gelatins set correctly. Chefs and home cooks utilize ice baths set at or below 32°F to chill liquids rapidly, ensuring the desired texture is achieved. In material sciences, understanding the behavior of materials at temperatures around 32°F is essential for selecting appropriate materials for construction and manufacturing in cold climates, ensuring durability and preventing structural damage.
FAQ Section
Why is the freezing point of water important in meteorology?
The freezing point of water is crucial for predicting frost, freezing rain, sleet, and snow, which are vital for weather forecasting and ensuring public safety and infrastructure management.
How does understanding the freezing point help in cooking?
In cooking, particularly for desserts, knowing the freezing point ensures that ingredients achieve the right texture, such as setting ice cream and sorbets properly.
By grasping the concept of the freezing point in Fahrenheit, professionals and enthusiasts alike can better navigate the intricate interplay between temperature and various practical applications, ensuring efficiency, safety, and quality in their respective fields.