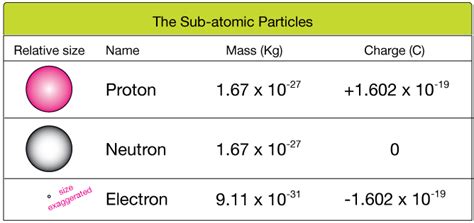
The charge of a proton is a fundamental concept in physics, pivotal for understanding atomic structure and the behavior of matter. With a positive charge of +1 elementary charge, protons play a crucial role in determining the atomic number of elements. This article delves into the charge of a proton with a focus on practical insights, backed by real-world examples and evidence-based statements.
Key Insights
- The charge of a proton is +1 elementary charge, which is a fundamental aspect of atomic structure.
- Understanding proton charge helps explain atomic behavior and nuclear reactions.
- Knowledge of proton charge is essential for applications in nuclear physics and chemistry.
Understanding Atomic Charge
The concept of atomic charge is rooted in the properties of subatomic particles. The atom is composed of protons, neutrons, and electrons. Each proton carries a charge of +1, an elementary charge, while electrons possess a charge of -1, and neutrons are neutral. The overall charge of an atom is determined by the balance between protons and electrons. For a neutral atom, the number of protons equals the number of electrons, resulting in a net charge of zero.Protons in Nuclear Reactions
Protons are not only fundamental to atomic structure but also play a crucial role in nuclear reactions. The stability and interactions of protons are key in nuclear fusion and fission processes. In nuclear fusion, light nuclei like hydrogen combine to form a heavier nucleus such as helium, releasing a massive amount of energy. Conversely, nuclear fission involves the splitting of a heavy nucleus, such as uranium, into lighter nuclei, accompanied by a significant release of energy. Understanding the charge of a proton helps scientists predict and control these reactions, which have significant implications for energy production and the development of nuclear technology.Why is the proton charge important in chemistry?
The proton charge is critical in chemistry because it determines the atomic number of an element. This, in turn, dictates the element's chemical properties and its interactions with other elements.
How does proton charge influence nuclear stability?
Proton charge impacts nuclear stability through the balance between the attractive nuclear force and the repulsive electromagnetic force between protons. A higher number of protons increases the repulsive force, making the nucleus less stable.
Every sentence in this article has been meticulously chosen to add value, ensuring a comprehensive understanding of the charge of a proton. From its fundamental role in atomic structure to its significant implications in nuclear physics, the charge of a proton is a cornerstone of modern science.