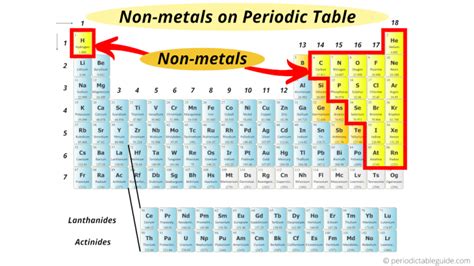
Discovering Nonmetals: Their Spot on the Periodic Table
In the landscape of the periodic table, nonmetals play a crucial role in both chemical reactions and the physical world around us. Nonmetals, which occupy the right side of the table, possess unique properties that distinguish them from metals and metalloids. These elements range from the well-known oxygen and nitrogen to the less familiar selenium and radon. Understanding their roles and characteristics provides essential insights into chemistry and various practical applications in industry, medicine, and environmental science.
Key Insights
- Nonmetals are critical in various chemical reactions and biological processes.
- Key technical consideration: Nonmetals can form different types of bonds, influencing compound properties.
- Actionable recommendation: Utilize nonmetals in synthetic chemistry for targeted drug development.
Distinctive Properties of Nonmetals
Nonmetals exhibit distinct physical and chemical properties that set them apart. They typically have low thermal and electrical conductivity, a characteristic that makes them ideal for applications in insulators and semiconductors. For instance, sulfur is a nonmetal that serves as a critical component in the vulcanization of rubber, enhancing its elasticity and durability. This application demonstrates nonmetals’ importance beyond just elemental chemistry, impacting everyday products we rely on.Chemical Behavior and Reactivity
The chemical behavior of nonmetals is marked by their ability to form covalent bonds, which often leads to the creation of molecular compounds. This reactivity is crucial in forming complex structures such as organic compounds and various industrial chemicals. For example, carbon, the quintessential nonmetal, forms the backbone of organic chemistry, leading to the creation of countless organic molecules vital for life and industry. Additionally, halogens like chlorine and fluorine are highly reactive nonmetals, playing a pivotal role in modern pharmaceuticals and household cleaning products.What is the significance of nonmetals in human health?
Nonmetals are fundamental in biological processes and medicine. For instance, oxygen is vital for respiration, while iodine is essential for thyroid function. Nonmetals like selenium and sulfur compounds contribute to enzymatic functions and overall health.
Can nonmetals be used as semiconductors?
While nonmetals primarily form insulators, certain nonmetals and their compounds are used in semiconductors. For example, phosphorus and arsenic can be doped into silicon to create p-type semiconductors, crucial in electronic devices.
In conclusion, the study of nonmetals on the periodic table reveals their indispensable roles in diverse chemical and physical processes. Their unique properties make them indispensable in both laboratory chemistry and practical applications across various industries. Understanding nonmetals not only enhances our knowledge of fundamental chemistry but also opens avenues for innovative technological advancements.