Welcome to your comprehensive guide on zero order kinetics, an often overlooked yet incredibly important concept in chemical reactions. If you’ve ever wondered why certain reactions proceed at a consistent rate, regardless of the concentration of reactants, this guide is for you. This detailed walkthrough will simplify the complex world of zero order kinetics, offering you real-world examples, actionable advice, and easy-to-follow solutions to help you grasp this fundamental principle in chemistry.
Understanding the Problem: Why Zero Order Kinetics Matters
In many chemical reactions, the rate is determined by the concentration of the reactants, following first or second-order kinetics. However, there are situations where the reaction rate remains constant, regardless of the concentration of the reactants. This phenomenon is known as zero order kinetics and it plays a crucial role in various industrial and biological processes. For instance, enzymes often work best at a specific concentration where the reaction rate is not dependent on substrate concentration anymore, leading to zero order kinetics. Understanding this concept is vital for optimizing chemical processes and ensuring efficient, consistent results.
Quick Reference Guide
Quick Reference
- Immediate action item: If you observe a reaction where the rate remains unchanged with varying concentrations, it’s likely zero order kinetics at play.
- Essential tip: For zero order reactions, the rate law is expressed as Rate = k, where k is a constant, indicating that the reaction rate is independent of reactant concentration.
- Common mistake to avoid: Confusing zero order with first or second-order kinetics, where reaction rates depend on reactant concentrations.
Detailed How-To: Understanding Zero Order Reactions
To thoroughly understand zero order kinetics, let’s break down what it entails, how it can be identified, and how it differs from other orders of kinetics. We’ll start with the basics and progressively move to more advanced concepts.
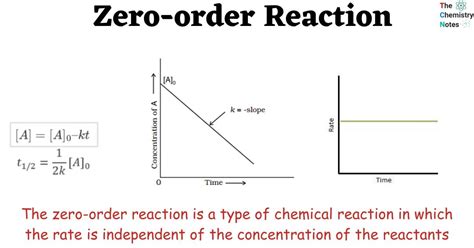
What is Zero Order Kinetics? Zero order kinetics refers to a reaction where the rate of the reaction remains constant, regardless of the concentration of the reactants. The rate law for zero order reactions can be written as:
Rate = k
where 'k' is the rate constant. Since the rate does not depend on the concentration of the reactants, the units of k in zero order reactions are concentration/time.
Identifying Zero Order Kinetics: To determine if a reaction follows zero order kinetics, you can analyze the concentration vs. time data. Here’s a step-by-step process:
- Collect Data: Measure the concentration of the reactant at different times.
- Plot the Data: Plot concentration vs. time. For zero order reactions, this plot will yield a straight line.
- Analyze the Graph: If the graph is a straight line, the reaction is zero order. The slope of the line gives the rate constant 'k'.
For instance, in enzymatic reactions at very high substrate concentrations, the enzyme works at its maximum rate, leading to zero order kinetics. This is often referred to as the Michaelis-Menten kinetics, where Vmax is the maximum rate, and it’s constant irrespective of substrate concentration.
Differences from First and Second-Order Kinetics: In first and second-order kinetics, the reaction rate is directly proportional to the concentration of one or two reactants, respectively. For first-order reactions, the rate law is Rate = k[A], and for second-order reactions, Rate = k[A][B]. To differentiate:
- First-Order: Plot ln([A]) vs. time. For a first-order reaction, this will yield a straight line with a slope of -k.
- Second-Order: Plot 1/[A] vs. time. For a second-order reaction, this will result in a straight line.
In contrast, the concentration vs. time graph for zero order reactions remains a straight line, unlike the curved lines in first or second-order reactions.
Applications in Industry: Zero order kinetics is prevalent in industrial processes where maintaining a constant reaction rate is crucial. For example, in large-scale production of chemicals, zero order reactions ensure consistent product quality. Similarly, in catalytic processes where catalysts reach saturation, leading to zero order kinetics, maintaining steady-state conditions is essential.
Practical FAQ Section
What are some common examples of zero order kinetics?
Zero order kinetics can be commonly observed in enzymatic reactions at high substrate concentrations and in catalytic reactions where the catalyst becomes saturated. Another example is the decomposition of a chemical compound where the rate of decomposition is constant due to other factors controlling the rate, such as temperature. Additionally, in photochemical reactions, when the light intensity is so high that it saturates the reaction, zero order kinetics can be observed.
How can I determine if a reaction follows zero order kinetics experimentally?
To determine if a reaction is zero order experimentally, you need to plot the concentration of the reactant versus time. If the plot is a straight line, it indicates zero order kinetics. The rate constant ‘k’ can be determined from the slope of this line. Alternatively, if you have data showing that the rate of reaction is constant regardless of changes in the concentration of reactants, you can conclude it’s zero order. Conducting experiments at varying concentrations but holding other variables constant, such as temperature, can provide further insight.
Why is zero order kinetics important in drug metabolism?
In drug metabolism, zero order kinetics is crucial because it means that the rate of drug elimination from the body is constant, irrespective of the drug concentration. This is important for understanding drug dosing regimens. If a drug exhibits zero order kinetics, small changes in drug concentration can lead to large changes in the amount of drug remaining in the body. Understanding this helps in developing dosage schedules that ensure the drug remains at therapeutic levels without reaching toxic levels.
This guide aims to demystify zero order kinetics by providing a clear, actionable, and detailed understanding. From identifying zero order reactions to understanding their practical applications, you now have a solid foundation to apply this knowledge in real-world scenarios. Whether you’re a student, a professional, or simply curious about chemical reactions, this guide equips you with the necessary tools to navigate and leverage zero order kinetics effectively.