Understanding the Bohr Atomic Model is essential for anyone interested in chemistry, physics, or even a basic grasp of how matter works at its most fundamental level. This guide is designed to help you navigate the complexities of carbon’s orbital structure using Niels Bohr’s model of the atom. With step-by-step guidance and practical examples, we’ll cover the essentials to make this intricate subject accessible.
Whether you're a student, a professional scientist, or just someone with a keen interest in the microscopic world, this guide will help you decode carbon's orbital secrets. By addressing common pain points and providing actionable advice, we aim to transform your understanding of the Bohr atomic model, particularly as it applies to carbon atoms.
The Need for Understanding the Bohr Atomic Model
The Bohr atomic model provides a visual and mathematical representation of an atom, emphasizing the arrangement of electrons in orbits around the nucleus. For carbon, understanding its electron configuration helps explain its chemical properties and behavior in various reactions. By comprehending the Bohr model, you gain insights into not just carbon, but all elements and their reactions.
However, many find the Bohr model challenging due to its abstract nature and the intricate details it involves. The complexity can seem overwhelming, especially when delving into elements like carbon, which has six electrons requiring specific configurations. This guide aims to demystify the Bohr model by breaking down these complexities into manageable and understandable parts, making it easier for you to grasp the fundamentals and apply them practically.
Quick Reference
Quick Reference
- Immediate action item: Sketch the Bohr diagram for a carbon atom to visualize electron placement.
- Essential tip: Remember, electrons fill the lowest energy level first before moving to higher levels.
- Common mistake to avoid: Confusing electron shells with energy levels; ensure clarity between the terms.
Step-by-Step Guide to Carbon’s Orbital Configuration
Let’s embark on a journey through carbon’s electron configuration. We’ll break it down into digestible parts, ensuring you understand each layer of complexity. The Bohr model, with its concentric circles representing electron shells, is a powerful tool for visualizing this configuration.
Understanding Electron Shells
In the Bohr model, electrons occupy discrete energy levels or shells. For carbon, we start with the first two shells:
- The first shell (K-shell) can hold up to 2 electrons.
- The second shell (L-shell) can hold up to 8 electrons.
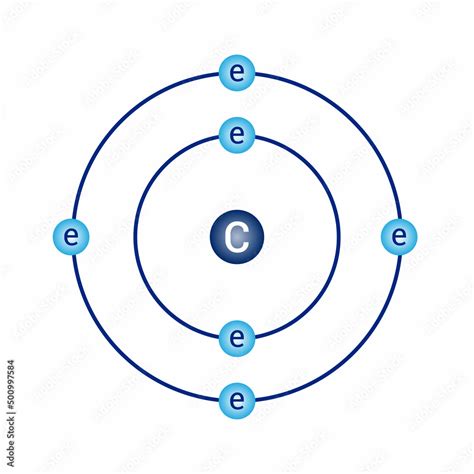
Given carbon’s atomic number of 6, it has 6 electrons to distribute across these shells.
Filling the Shells According to Bohr’s Model
Bohr proposed that electrons orbit the nucleus in fixed paths or shells with specific energy levels. Carbon’s electrons are distributed as follows:
- Two electrons in the first shell (K-shell)
- Four electrons in the second shell (L-shell)
This configuration places carbon in Group 14 of the periodic table, explaining its chemical properties.
Visualizing Carbon’s Electron Configuration
To get a visual grasp of how carbon’s electrons are arranged:
- Draw a circle to represent the nucleus.
- Draw a smaller circle around the nucleus to represent the first electron shell (K-shell), and place two electrons here.
- Draw a larger circle around the nucleus to represent the second electron shell (L-shell), and place four electrons here.
Practical Application: Why This Matters
Understanding carbon’s electron configuration isn’t just academic. It’s foundational for comprehending organic chemistry, the basis of life on Earth. Carbon’s ability to form four covalent bonds makes it the cornerstone of organic molecules, from DNA to plastics.
Advanced Understanding: Carbon’s Orbital Energies
While Bohr’s model uses shells to describe electron energies, a more advanced view uses orbitals. Carbon’s 2s and 2p orbitals are critical in its chemical interactions. Here’s a deeper dive:
- The 2s orbital can hold two electrons and is lower in energy than the 2p orbitals.
- The 2p orbitals are split into three orbitals, each capable of holding two electrons, oriented differently in space.
Carbon’s electrons fill the 2s orbital first, then distribute among the three 2p orbitals.
Practical Insight: Why Orbitals Matter
Orbitals provide a more accurate depiction of electron positions and energies. Understanding orbital configurations helps predict chemical reactivity and bonding patterns.
Practical FAQ
What is the significance of carbon’s electron configuration in chemical reactions?
Carbon’s electron configuration is pivotal because it dictates how carbon atoms interact with other atoms. With four valence electrons, carbon can form four covalent bonds with other atoms, creating a vast array of compounds. This ability to form stable bonds with a variety of elements is why carbon is so central to life and materials science. Understanding this configuration helps predict how carbon will behave in different chemical environments, making it a key concept for anyone studying chemistry.
Common Pitfalls and How to Avoid Them
One common mistake is assuming all orbitals are equally likely to be filled. In reality, electrons fill the 2s orbital before the 2p orbitals due to lower energy levels. Another error is confusing the concept of electron shells with orbitals, leading to misconceptions about electron distribution.
To avoid these pitfalls, always remember:
- Electron filling order: Follow the Aufbau principle – electrons fill the lowest available energy levels first.
- Correct terminology: Use “shells” for energy levels and “orbitals” for the space where electrons are likely to be found.
Conclusion: Embracing the Bohr Model
The Bohr atomic model might seem daunting at first, but with a clear understanding of its principles, you can unravel the mysteries of carbon’s orbital configuration. This journey through electron shells and orbitals equips you with a foundational understanding of atomic structure and chemistry. By embracing this model, you open the door to a deeper appreciation of the microscopic world and its impact on the macroscopic world we live in.
Remember, the goal is not just to memorize facts but to understand the principles that govern atomic behavior. With this guide, you’ve taken a significant step toward mastering the Bohr atomic model, particularly as it relates to carbon. Keep practicing, exploring, and applying these concepts to fully grasp their implications in chemistry and beyond.
This guide has provided a comprehensive introduction to the Bohr atomic model, focusing on carbon’s orbital configuration. It is designed to be practical, addressing user pain points while offering actionable advice and clear progression from basic to advanced understanding.